@SignantHealth
n recent years, we have seen enhanced emphasis on the importance of patient-reported outcome measures (PROM) collected in oncology clinical trials reflected by the FDA. Project Patient Voice, for example, was established to provide a mechanism to share patient-reported symptom data from cancer clinical trials with approved treatments to provide more information to patients and healthcare providers in treatment decision making.
These are very reasonable concerns. In terms of specificity of common measures, Gnanasakthy et al. report that common generic and disease-specific scales used in oncology trials, such as those developed by the European Organisation for Research and Treatment of Cancer (EORTC) and Functional Assessment of Chronic Illness Therapy (FACIT), may lack sensitivity and specificity in several ways. Some scale items may be irrelevant in certain circumstances, some may not be precise enough to capture the patient experience with specific therapies, and summary scores for total and subscales may be insufficiently sensitive due to equal weighting of items leading to dilution of the impact of the most meaningful symptoms. Further, common subscales developed within existing PROMs may not be specific enough to the measurement of single domains of interest and therefore lead to less robust conclusions.
In oncology trials, patients are typically asked to complete PROMs at clinic visits at the start of each cycle of treatment. This is the point at which patients have recovered sufficiently from the previous treatment to receive the next cycle. If patients are not well enough to commence treatment, then cycle starts and associated PROM assessments are delayed. This assessment schedule is therefore less optimal for PROMs assessing the impact of treatment.
The FDA’s recent draft guidance on core patient-reported outcomes (PROs) in cancer clinical trials proposes an approach to addressing a number of these concerns and will enable robust and reliable PROM data for better regulatory consideration and potentially increased inclusion of this data on US oncology drug labeling.
We reviewed the draft guidance and its impact on the selection and implementation of PROMs in oncology clinical trials, to provide recommendations for future study planning.
Key Elements of the Draft Guidance
Building from earlier published work, the FDA identifies a core set of PROMs to separately measure the following concepts:
- Disease-related symptoms
- Symptomatic adverse events (AEs)
- Overall side effect impact measure (single item)
- Physical function
- Role function
Standard instruments exist and are commonly used for some of these concepts.
Disease-Related Symptoms
The draft guidance cites only one example of a disease-related symptom measure: the Critical Path Institute’s NSCLC-SAQ for non-small cell lung cancer. This may correspond to the commentary of Gnanasakthy et al. in terms of their considerations of the possible limitations of common scales such as those provided by EORTC and FACIT. They conclude that these scales may contain some irrelevant items when used in certain patient groups, may not consistently assess the most important symptoms, and may provide subscores that are diluted by applying equal weight to less important symptoms.
It’s important to note that the requirements of the FDA draft guidance are to be able to report measures of each of the five domains separately. Some existing instruments and their subscale scores may not be immediately suitable for this in their current form. For example, in the FACT-B measure, commonly used in breast cancer, items within the “physical well-being” domain include both treatment-related symptoms (e.g., “I am bothered by side effects of treatment”) and disease-related symptoms (e.g., “I have pain”). The causality in changes in the subscale measure, therefore, cannot be easily distinguished between treatment-related or disease-related symptoms. However, it is possible that new subscales could be developed from these existing instruments to meet the required specificity of the FDA requirements. This could also be achieved by selecting pertinent items from emerging item libraries, such as that developed by EORTC. In both cases, with scale author approval, additional content validity and other work may be required to support the new subscale measures.
Symptomatic Adverse Events (AEs)
The Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE) is well accepted for the collection of symptomatic AEs and is cited in the draft guidance as an example instrument. This tool uses an item library from which specific AE items are selected for assessment, and sponsors should provide a careful rationale to support the selection of a concise set of the most important and/or high-frequency AEs.
The draft guidance also suggests consideration of additional free text items to capture missing important symptom items. This does present a challenge for researchers, as free text data is harder to manage and analyze than items with defined response options. While electronic solutions can collect free text data, key data management considerations include how to deal with ambiguous or misspelled entries and how to manage the different languages used.
It’s important to note that while PRO-CTCAE data are reported and analyzed independently of clinician-reported AEs, these self-reports can provide a valuable input to inform and enhance clinician identification, documentation, and reporting of adverse events. The self-report data without clinician interpretation does not fall within the requirements of the formal safety reporting processes associated with clinician-reported AEs, but timely review of these data alongside clinician assessment may enhance the detection and interpretation of AEs by the investigator.
Overall Side Effect Impact Measure
We rarely see the inclusion of an independent single item to summarize the overall impact of side effects in today’s oncology protocols. A global measure of side effect impact is a valuable enhancement, as it includes the impact of any side effects the patient may experience that were not measured using the PRO-CTCAE implementation, and the additional insight enables patients to attach greater importance to certain side effects when determining the collective impact of all side effects experienced. Tools already exist for the measurement of overall side effect impact, including the use of a patient global impression of severity scale (e.g., “Please select the response that best describes the severity of your overall side effects from treatment over the past 7 days (where 0 represents none and 3 represents severe)”); the GP5 question from the FACIT item library (a five-point ordinal scale measuring the degree a patient is bothered by symptoms from “not at all” to “very much”); and the Q168 question from the EORTC item library (a four-point ordinal scale rated from “not at all” to “very much”). These three approaches are cited within the draft guidance as examples of measures for overall side effect impact.
Physical Function
Physical function includes aspects such as walking, lifting, and reaching that are considered important for independent functioning. The draft guidance provides examples of measures of physical function, including the Patient-Reported Outcomes Measurement Information System (PROMIS) physical function item library and EORTC QLQ-C30 physical function subscale. Some authors consider the PROMIS scale to have benefits over the EORTC subscale because it may be more versatile and support a wide range of severities and ages of patients. For example, the EORTC subscale items use terms such as “strenuous” or “limited doing” which may be interpreted contextually differently by older patients or those with more advanced disease. An advantage of the EORTC instrument, however, is that it is used to measure other constructs in addition to physical function.
Role Function
In this context, role function should measure the impact of a treatment on the ability to work and carry out daily activities. The draft guidance cites the EORTC QLQ-C30 role function subscale as an example measure.
Frequency of Administration
The draft guidance recommends more frequent assessment in early cycles and fewer later in the treatment process; this means at-home measurements in addition to (or instead of) on-site visits. We acknowledge the importance of this approach to capture the full impact of the disease and its treatment. It is common current practice in oncology trials to measure PROs at clinic visits at the start of each cycle of treatment. This is the point at which patients have recovered sufficiently from the previous treatment to receive the next cycle. If patients are not well enough to commence treatment, then the next cycle and associated PROM assessments are delayed. There is an aspect of convenience in this, as these PROMs can be measured during clinic attendance and do not require at-home completion solutions. There is also a perception that patients may be unwilling or unable to complete PROMs during the earlier stages of each cycle due to the debilitating effects of treatment. However, if we want to truly measure treatment-related symptoms, then it will be important to consider the optimal timing of these assessments to obtain a full and accurate picture. Our qualitative research on oncology patients confirms that the early stages of a cycle are debilitating, but despite this, patients are willing to provide data.
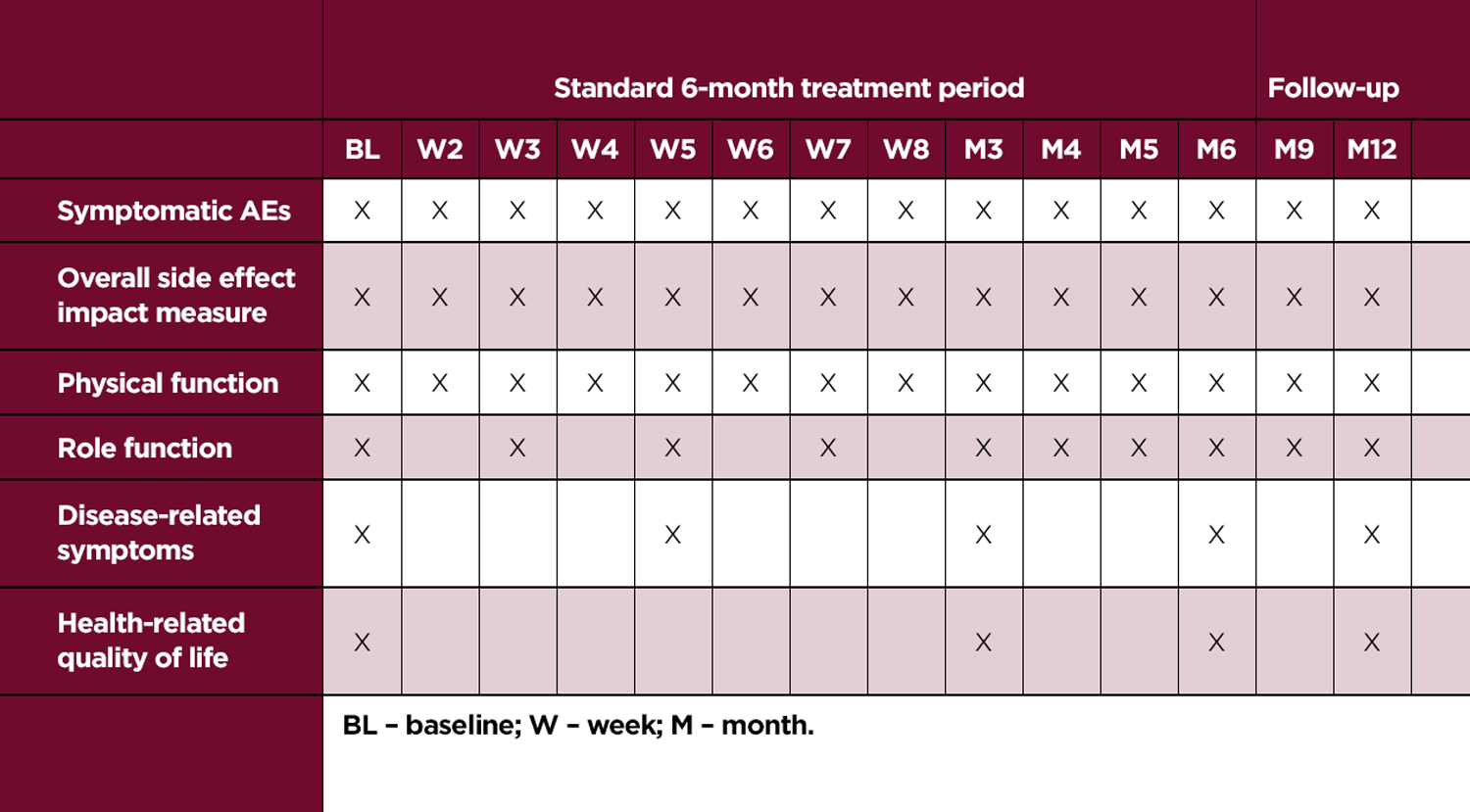
In their example schedule of assessment (Figure 1), the FDA illustrates the more frequent measurement recommended during the earlier part of the trial. The example suggests weekly measurement of symptomatic AEs, the overall impact of side effects item, and physical function for the first eight weeks before moving to a monthly cadence and then quarterly as patients progress through the 12-month treatment period. This is a change from the current pre-cycle measurement approaches we commonly see, requiring patients to complete instruments at-home during the early cycles of treatment, and optimizing measurements in the trial period when most patients are enrolled.
Considerations and Recommendations for Future Trial Designs
Implement a solution for the collection of PROMs in the at-home setting. This should be used to enable the higher frequency of assessments for certain measures earlier in the treatment period and should contain features to help drive timely entries and complete datasets. Less frequently measured PROMs could be collected at home or on site depending on the visit schedule and could enable flexibility in administration setting. Site-based collection may be associated with higher completion rates when measurements are less frequent.
Measurement Selection
To meet the requirements of the FDA draft guidance, PROMs should enable the discrete measurement of symptomatic AEs, overall side effect impact, physical function, role function, and disease-related symptoms. It will be important to select PROMs that contain appropriate means of measuring and reporting these domains independently and specifically.
PROM Development/Adaptation
In some cases, existing instruments or item libraries may require additional work to identify the correct set of items to measure each domain specifically and comprehensively for the disease studied. This may require discussion with scale authors to apply adaptations to subscores, or to develop pertinent item sets from libraries, and conduct any associated content validity and psychometric evaluation.
Mitigate Missing Data
Ensuring that the measurement strategy is in line with FDA requirements is important, but attention must also be given to the FDA concern that missing data may limit the ability to draw robust conclusions. Attention to site and patient training, and ensuring that the solution to collect PROM data includes methods to remind and proactively monitor PROM completion, are important considerations in limiting missing data and drawing reliable conclusions which may lead to consideration for labeling claims. Electronic solutions are well suited to these requirements. Solutions should also capture reasons for missing assessments and enable PROMs to be collected at the point of withdrawal, where appropriate.
As an industry, we should welcome this draft guidance. It provides clear detail of the core domains that are of most value to the agency in their review of PROM data in new oncology drug applications. Their attention to measurement frequency and missing data ensures that data collected can be used to derive reliable clinical endpoints. While it remains to be seen whether this will ultimately translate into more PROM-based data being included on approved US oncology drug labeling, we must assume that this guidance represents a positive signal.

