AU Consulting
PharmaDJ
t2T Healthcare, Inc.
hina and Japan, two large economies in Asia, have shared a long and intricate history of cultural exchange and mutual influence dating back to ancient times.
Historic and Parallel Changes in Drug Development
Japan’s capability in drug development earned it a seat on the International Council for Harmonisation (ICH), established in 1990, as one of the founding members alongside the European Union and the United States. The country aligned its clinical trials with international standards when its health authority (the Ministry of Health, Labour and Welfare [MHLW]) adopted the ICH GCP Guideline in the Ministerial Ordinance in 1997. Later on, the country’s policy to enhance participation in multiregional clinical trials (MRCTs) was articulated in the MHLW Notification Basic Principles on Global Clinical Trials (2007).
China’s pharmaceutical regulatory system was different from that employed in Western countries. However, a pivotal shift began in 2015 with the State Council’s statement Reform of the Drug and Medical Device Evaluation and Approval System (Guo Ban Fa No. 44), which initiated the internationalization and harmonization of drug regulation in China. The State Council statement clearly described China’s intent to modernize its drug registration system by introducing new ideas such as a definition of “new drug,” accelerating reviews of NDAs/INDs (New Drug Applications/Investigational New Drug applications), and regulatory harmonization. Other key milestones quickly followed, including China’s formal accession to ICH in 2017, which visibly reflected the presence of China in the international pharmaceutical ecosystem; the Opinions on Deepening the Reform of Review & Approval System to Encourage Innovation in Drugs and Medical Devices (No. 42), which described a concrete plan to update drug regulations, also issued in 2017; and the 2019 Drug Administration Law Revision, a full revision of the pharmaceutical law to complete the evolutionary innovation of the drug registration system in China. China accomplished in three to four years what took Japan nearly 20 years to achieve.
Iyaku Kenpatsu Tatsujin (Yiyao Yanfa Daren in Chinese, Pharmaceutical R&D Experts in English) Laid the Groundwork for Bilateral Communication
This internationalization has created an environment where people in both nations can speak a common language with regard to drug regulation.
The authors started working together to launch an initiative in July 2021: the online media outlet Iyaku Kenpatsu Tatsujin (Pharmaceutical R&D Experts) was designed to help pharmaceutical industry information flow between Japan and China. Articles and updates were published twice a month until 2024 on a website run by the Chinese biopharmaceutical industry media outlet PharmaDJ (which supported this activity by providing their platform) and were also posted on the popular Chinese social networking service/site WeChat. The articles in this initial phase were mostly about Chinese pharma industry news to be provided to the Japanese industry in the Japanese language.
The Japanese edition of Tatsujin was published until March 2024, when PharmaDJ changed its position to temporarily stop website support for this activity. However, right after that, interest had been sparked and in response to growing demand from rapidly internationalizing Chinese drug firms, a Chinese edition of Tatsujin, called “Yiyao Yanfa Daren: Chuhai Riben” in Chinese (in English: Pharmaceutical R&D Experts: Go Abroad to Japan), was launched in February 2025. This Chinese edition provides Japan-related industry information in the Chinese language for Chinese readers, printed and delivered by mail twice a month, continuing this grassroots advocacy for communication and friendship between these two countries. There is no Japanese-language edition at this time.
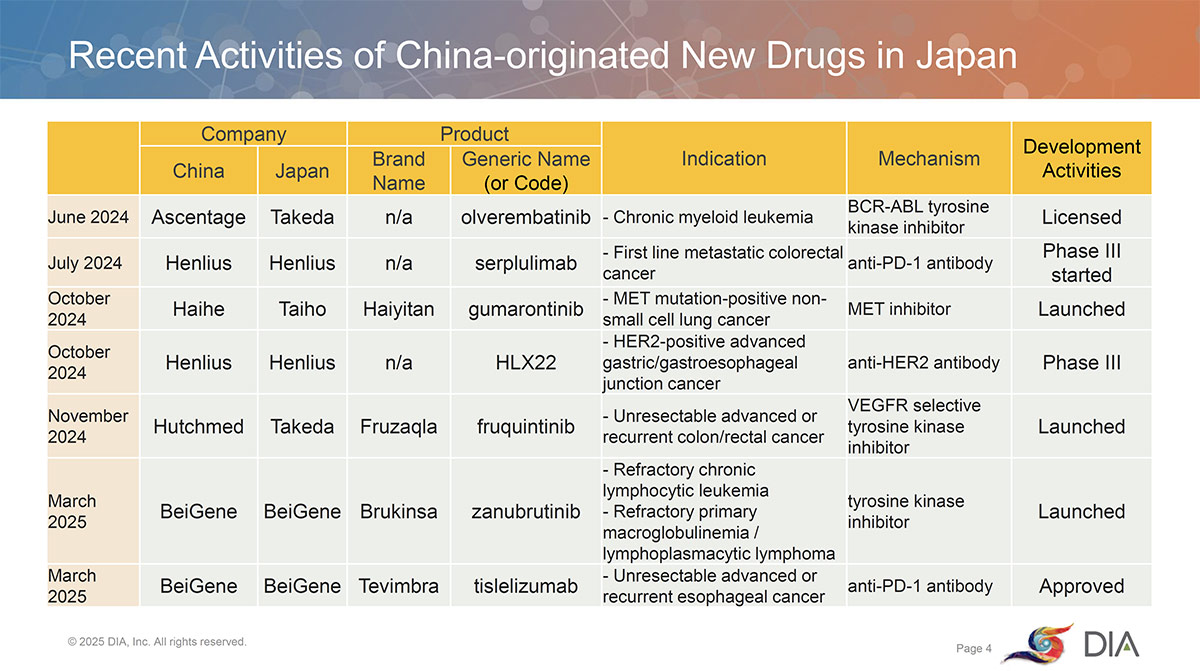
Sessions Reflect the Growth of Chinese Drug Companies
Sessions at two recent DIA China Annual Meetings (CAM) have also enhanced this bilateral relationship and reported on the remarkable growth of Chinese drug companies as well as their engagement in and with Japan.
The CAM 2023 session Japan as the Next Destination: Demystify the Japanese Regulations presented the results of a survey conducted by the authors in collaboration with the Pharmaceutical Innovation and Research Development Association (PhIRDA) of China. The association’s member companies were canvassed about their priorities regarding foreign countries in the context of the strategies to expand their market to other countries in the world (Figure 1).