Around the Globe
Re-Evaluating the Need for Comparative Clinical Studies
t’s been ten years since the enactment of the Biologics Price Competition and Innovation Act, which created a new approval pathway for biosimilar products in the US. This new pathway was touted as a critical way to provide broader access to cost-effective biological treatments and has contributed to 26 approved biosimilars for nine different reference products as of March 2020. While much has evolved since the legislation was introduced, what has remained relatively constant is the comparative clinical study (CCS) as a necessary gateway to support the approval of biosimilar products. However, with advances in analytical techniques, improved understanding of the relationship between protein structure and function, and an ever-increasing body of global biosimilar experience, it’s time to re-evaluate the consistent need for CCSs in the next decade of biosimilar development.
The First Decade: What Was the Purpose of a CCS?
Within this stepwise approach, FDA maintains authority to waive a CCS if it is deemed unnecessary to support approval based upon totality-of-the-evidence. However, multiple factors have continued to necessitate including a CCS as part of most biosimilar development programs. In retrospect, inclusion of a CCS has been beneficial because it generated data which helped engender patient and healthcare provider confidence in biosimilar products, and develop experience with this new regulatory approval framework in the US.
Trastuzumab: A CCS Case Example
Although the extrapolation of data for indications previously approved for a reference product remains a significant upside to biosimilar developers, challenges with conducting a CCS remain. For instance, it is estimated that a phase 3 clinical trial enrolling 600 patients can cost $50M US. Other factors like time investment (CCSs are often ≥ 1 year in duration) and recruitment (needing to share the same patient pool with novel therapeutic studies) could also create barriers for smaller companies to bring biosimilars to market.

** Study 5 only; Study 6 was a single arm trial
*** Study 5 (n=469) and Study 6 (n=222)
Exploring New Approaches
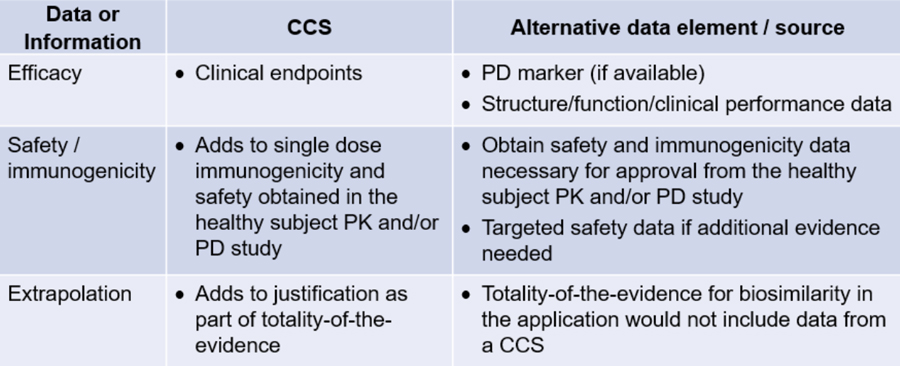

Potential Obstacles to Eliminating the CCS
Efficacy
Safety
Extrapolation
Scientific Flexibility
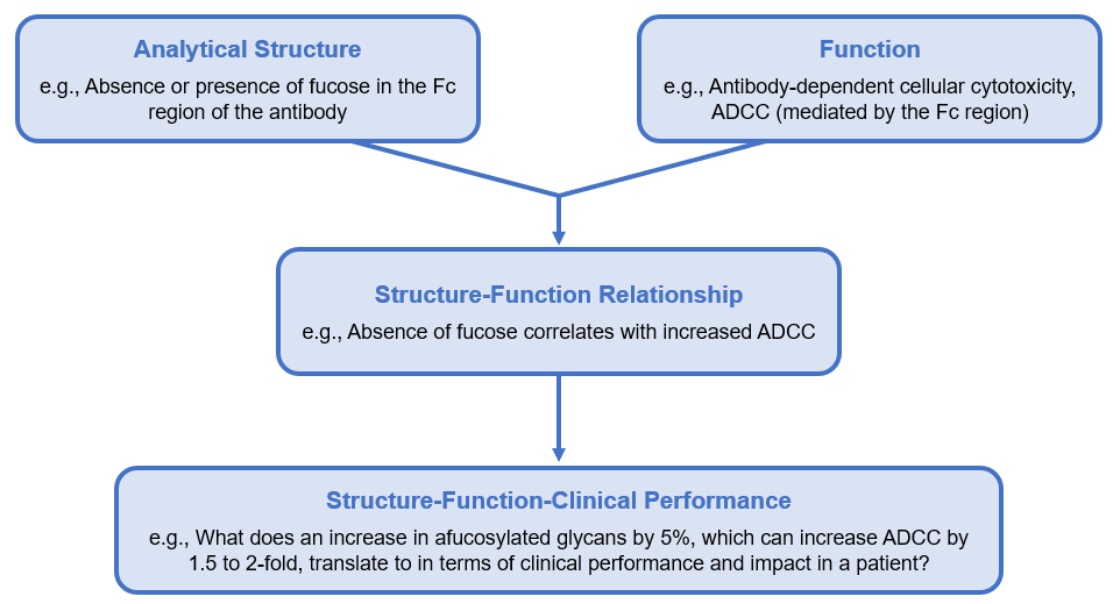
It may be many more years before technological advances, structure-function-clinical performance understanding, and global experience with biosimilars completely align to eliminate the necessity for CCS data. Until then, revisiting the current role of the CCS in light of knowledge and data gathered over the first ten years of US biosimilar experience should be considered in order to further advance the goals of expanded market access to critical biological treatments in the next decade.
References available upon request.

