Astellas Pharma Inc.
Peaceful Inc.
Nippon Medical School
National Center for Child Health and Development
Metropolitan Academic Research Consortium
Tokyo Medical University
Metropolitan Academic Research Consortium
Fortrea Japan K.K.
cademic institutions in Japan produce abundant life science research “seeds,” yet many of these seeds never blossom into practical pharmaceuticals for patients (see Cabinet Secretariat Action Plan 2024). This article presents a case study led by the DIA Japan Open Innovation Community (see previous article) that tested this hypothesis: Direct, in-person scientific exchange between academic and industry researchers—within the familiar setting of a corporate research facility—could dismantle communication barriers, foster collaboration, and accelerate the societal implementation of academic discoveries.
Japan’s Persistent Drug Lag and Drug Loss
Compared to the US, Japan’s drug discovery ecosystem continues to grapple with the persistent challenge of drug lag and drug loss (see this report [Japanese only] from the Ministry of Health, Labour and Welfare). Drug lag and drug loss in general means delay of commercialization or lack of development of drugs which have been approved outside Japan. However, in this article, drug lag and drug loss also means promising innovations originating in academia either never achieve commercialization or are significantly delayed in reaching the market.
While the cause of this lag/loss is generally related to high costs for clinical trials in Japan and regulatory requirements on Japanese data for submission to Japanese authorities, in reality it may be a structural problem occurring further upstream. More than one-fourth of drugs approved in the US are discovered by academia or startup companies, demonstrating a robust pathway from research to product. Although Japanese universities produce a wealth of high-quality basic research findings, these discoveries seldom advance to become approved medicines.
One underlying factor is the low mobility and limited interaction between academic and industry researchers. Cross-sector personnel exchanges are rare compared to the US, and researchers often remain within the confines of their own institutions. This isolation limits mutual understanding and reduces opportunities for collaboration in Japan.
Hypothesis
Creating a physical venue for direct, face-to-face interaction based on shared scientific language could stimulate collaboration and overcome these systemic barriers.
Methodology and Event Design
Collaborative Partnerships
To test this hypothesis, the DIA Japan Open Innovation Community partnered with the Metropolitan Academic Research Consortium (MARC) to represent the academic sector (mainly private medical universities in the Tokyo metropolitan area such as Keio University [Tokyo], Nippon Medical School [Tokyo], Tokyo Medical University, Saitama Medical University [Saitama], and Kitasato University [Kanagawa]) and with a global, Japan-headquartered pharmaceutical company to represent the industry.
The resulting symposium was hosted by the company’s Research Center Conference Rooms to intentionally invert the conventional collaborative model: To bring academia into industry’s physical space instead of asking industry researchers to leave their workplace to investigate academic research.
Program Structure
Session 1: Keynote Session: Representatives from academia, industry, and public agencies delivered talks on their perspectives regarding open innovation in drug development.
Session 2: Interactive Poster Session (Standing Buffet Style)
Poster presentations by researchers from academia, startups, and pharmaceutical companies on innovative projects (including AI-driven drug discovery, new therapeutic candidates, and diverse approaches to advancing drug development and therapeutic strategies) were organized alongside catered refreshments, allowing participants to mingle freely, move between displays, and engage in spontaneous conversations.
Results
Participation and Engagement
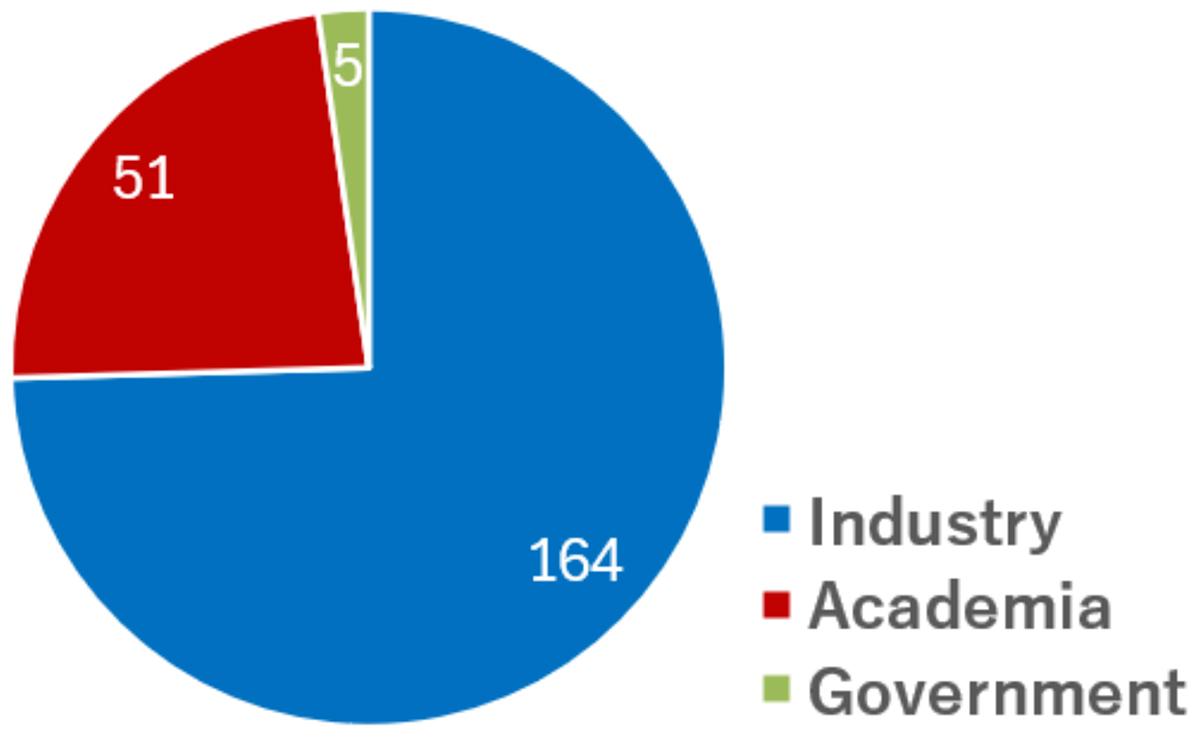
Attendance: 164 industry researchers, 51 academic researchers, and 5 government officials—220 total—including representatives of the Ministry of Economy, Trade and Industry (METI) and Japan Agency for Medical Research and Development (AMED).
Diversity: Academic scientists, industry researchers, and other multidisciplinary participants from university research administration, start ups, venture capital firms, etc.

- ≥80% rated all keynote presentations as “very beneficial”
- Positive comments emphasized diversity of perspectives and practical insights
- 88.3% expressed strong interest in attending future events
- Poster session survey comments:
- “Built new professional human networks”
- “Spoke with many researchers of different backgrounds”
- “Gained new insights regarding his/her research.”
Qualitative Insights
In the selection of the venue, this symposium was the first attempt to prioritize access to pharmaceutical research facilities over convenient access to business districts. Participants expressed a high level of satisfaction with the symposium in their survey response. Many attendees were more engrossed in the discussions than in the buffet!
Participants ranked “building professional networks” (39/127 responses [31%]) and “opportunities to speak with diverse researchers” (36/127 responses [28%]) positively in their Session 2 survey feedback. Seventeen respondents explicitly stated that they “engaged in discussions leading to concrete next steps; planning specific entries for government-funded projects like AMED and initiating collaborative research discussions”: direct evidence that their interactions evolved from polite business card exchanges into meaningful scientific discourse.
Industry attendees further praised the event’s design, noting that the posters functioned as “large business cards” that facilitated immediate discussion and collaboration. One participant described their enthusiasm as feeling like “the start of the dawn of Japan’s open innovation.” This was indeed the symposium’s strategic intent.
It seems reasonable to infer from these outcomes that participants demonstrated a clear appetite for exchange when provided a trusted, familiar setting for discussion anchored by common scientific themes. The quantity and quality of engagement at this event strongly challenges the assumption that Japanese researchers are reluctant to collaborate outside their institutions often attributed to structural issues like low researcher mobility, as noted in MHLW reports.
Reframed Perspective
This single event reframed the perspective of the DIA Japan Open Innovation Community: The challenge facing pharmaceutical innovation is not a lack of willingness to collaborate but an absence of structured opportunities to collaborate in specific environments. The survey results indicated that hosting this event in a corporate research environment removed the psychological barrier for industry researchers to work in an unfamiliar environment, while academic researchers expressed appreciation for this rare opportunity to access industry’s work.
Other companies could replicate embedding academia within an industry venue and could form a scalable blueprint for other open innovation efforts in Japan.
Conclusion
The DIA Japan Open Innovation Community, MARC, and the pharmaceutical company confirmed through this symposium that a shared scientific environment fosters meaningful cross-sector collaboration. Attendees emphasized the potential to scale and sustain this format and thereby transform Japan’s approach to academia-industry partnerships and address drug lag, drug loss, and other chronic challenges.

