How Africa’s New Medicines Pilot is Transforming Access to Safe and Effective Treatments
African Union Development Agency – New Partnership for Africa’s Development (AUDA-NEPAD)
Consultant to the Gates Foundation
magine being able to access life-saving medicinal products faster, no matter where you live on the African continent. This is the promise behind the Continental List of Human Medicinal Products Pilot, a landmark pilot program led by the African Union Development Agency – New Partnership for Africa’s Development (AUDA-NEPAD) through the African Medicines Regulatory Harmonization (AMRH) initiative.
What Is the Pilot About?
At its heart, the Continental List is about trust and transparency. It enables a continent-wide panel of regulatory expert assessors and inspectors from various African national regulatory authorities (NRAs) in countries such as Nigeria, Ghana, Senegal, South Africa, Tanzania, Kenya, and Rwanda, amongst others, to jointly review and issue a “Positive Opinion” on priority human medicinal products, ranging from essential vaccines to advanced cancer therapies.

This listing empowers national regulators across Africa to rely on the shared evaluation to accelerate their own approvals, thereby cutting down on duplication, shortening timelines, and reducing barriers to access.
The listings are not marketing authorizations (which refer to the official approval granted by an NRA allowing a medicine to be legally sold and distributed in a specific country) per se but are intended to be used by NRAs as the scientific basis for accelerated decision-making through reliance or recognition mechanisms. This model supports faster and more efficient access to essential medicines while promoting convergence in regulatory standards and capacity development.
Major Milestones and Achievements
Since its launch, the initiative has scored several historic achievements, including:
First-Ever Continental Listings (March 2025)
Five life-saving medicinal products were assessed and granted positive opinions by the AMRH Evaluation of Medicinal Products Technical Committee (EMP-TC) and approved for continental listing in the AMRH Green Book by the AMRH Steering Committee:
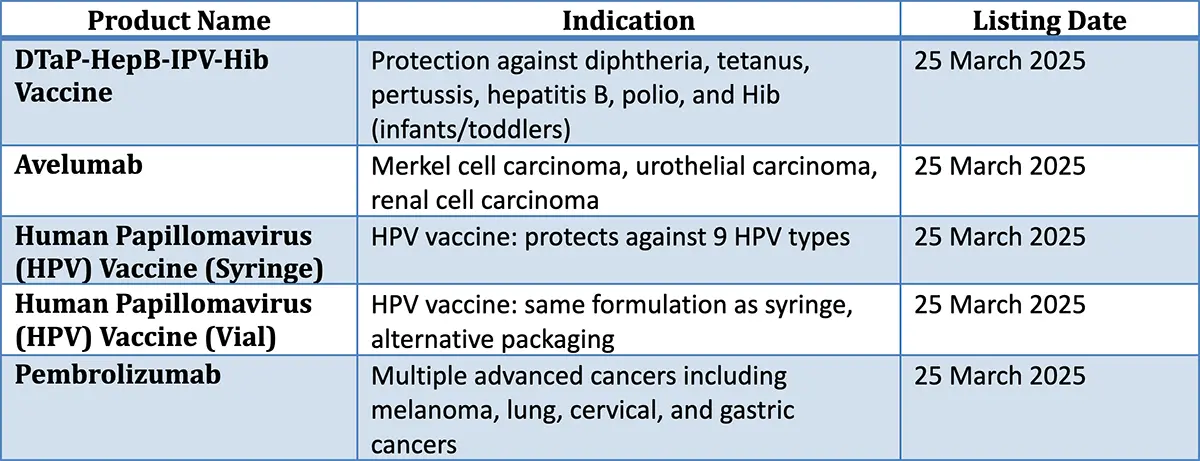
To date, ten (10) NRAs have authorized the listed medicines. The number of products submitted/number of products authorized: Botswana (1/5), United Republic of Tanzania (4/4), Ghana (3/3), Zambia (2/3), Zimbabwe (2/3), Kenya (1/2), Angola (1/1), Mozambique (1/1), Democratic Republic of Congo (1/1), and Ethiopia (5/5). It is important to note that not all products were submitted to all countries, and that dossier submissions at the country level are still ongoing. However, the continental listing enabled these innovative products to be available in four (4) African Regional Economic Communities (RECs) in under three (3) months, with some approvals being issued as early as two (2) weeks after the listing. This forms a stark contrast to the 24- to 48-month product approval timeline in some of these countries.
Strategic Value and Regulatory Impact
The Continental Listing embodies an African shift toward structured regulatory reliance, a key feature in global regulatory systems strengthening. By leveraging shared assessments and mutual trust, this joint assessment process enhances the regulatory maturity of participating countries and can be a useful tool for the African Medicines Agency (AMA).
This mechanism aligns with the WHO Global Benchmarking Tool (GBT) for regulatory system strengthening through reliance practice implementation and supports the African Union’s broader goals of achieving regulatory efficiency, regional coherence, and universal health coverage.
Why This Matters
African patients suffering from neglected tropical diseases (NTDs), such as schistosomiasis, onchocerciasis (river blindness), and trachoma, frequently face long delays in accessing life-saving medicines due to fragmented national regulatory processes and limited funding. NTDs have long been absent from the global health agenda and many NRAs are under-resourced, resulting in duplicative and inconsistent product reviews. As enhanced therapies for diseases like Lassa fever are currently in the clinical trial pipeline, with monoclonal antibody treatments (Arevirumab-3), antiviral agents (favipiravir), and other treatments (IAVI C105 vaccine, IMP-1088) showing early promise in this area, a robust regulatory mechanism is required to counter the disparate, uncoordinated country-specific assessments and authorizations of these critical medicines.
The Continental List offers that game-changing solution: one high-quality review accepted across multiple countries. In addition to reducing regulatory burden and improving decision consistency, ultimately helping to get safe and essential medicines to patients faster and more efficiently, innovative products that address African population diseases (such as NTDs) form part of the AMA’s specific mandate and the AMA will disseminate its opinion through this listing.
The Evaluation of Medicinal Products (EMP) and Good Manufacturing Practices (GMP) Technical Committees—composed of regulatory experts from across the continent, including the WHO maturity level 3 (ML3) NRAs [Tanzania Medicines & Medical Devices Authority (TMDA), Rwanda Food and Drugs Authority (Rwanda FDA), Egyptian Drug Authority (EDA), Ghana Food and Drugs Authority (Ghana FDA), Nigeria National Agency for Food and Drug Administration and Control (NAFDAC), Agence Sénégalaise de Réglementation Pharmaceutique (Senegal ARP), South African Health Products Regulatory Authority (SAHPRA), and Medicines Control Authority of Zimbabwe (MCAZ)]—played a vital role in ensuring the scientific integrity and consistency of the joint assessments during the pilot. These activities not only facilitated regulatory convergence but also contributed meaningfully to building the competency of NRA staff across participating countries. Capacity-building initiatives under the pilot further helped refine the pool of assessors, many of whom now form part of the emerging cadre of Continental Assessors, an important resource to be leveraged as the AMA becomes fully operational.
Progress to Date
- Efficient Timelines: The full product evaluation process took an average of 194-208 days, a significant improvement over traditional disparate country reviews, which can take 2-4 years.
- GMP Compliance: Manufacturing sites in India, Switzerland, Ireland, the US, and the Netherlands were assessed and found compliant with African GMP guidelines.
- Public Transparency: Each product listing includes a Public Assessment Report (PAR) and Summary of Product Characteristics (SmPC), available online to support national decision-making and to build stakeholder confidence.
The Road Ahead
The pilot laid critical groundwork for the AMA. The operationalization of the AMA is gathering speed with the appointment of its Board in 2024, and more recently in June 2025, that of the inaugural AMA Director General, Delese Mimi Darko, and as the AMA scales up its mandate and becomes operational, the Continental List mechanism will serve as a cornerstone for regulatory collaboration, efficiency, and convergence across Africa.
For patients, this means faster access to safe, effective treatments. For regulators, it represents a smarter, better coordinated system. And for Africa, it signals growing leadership in shaping global regulatory science.
To learn more about this topic, plan to attend our Middle East & Africa (MEA) Conference in Doha, Qatar.

