magine beginning a new treatment and not knowing if you’ll be able to manage the side effects—or if those side effects will be truly recognized/understood by your healthcare providers. Toxicities experienced over the course of a treatment (through both clinical trials and clinical practice) have real consequences as patients live with (and often quietly endure) the burdens and surprises that can accompany innovative therapies. In response to these challenges, there are new ways of incorporating patient perspectives into both drug development and the way treatments are assessed for approval.
Tolerability: A Function of Multiple Factors in a Patient’s Life
Tolerability goes beyond counting side effects; it is an ongoing negotiation between patients, their treatments, and their unique life situations. Novel long-term therapies can introduce new challenges for both patients and families, blurring the lines between disease symptoms, drug effects, and even ordinary aging. Financial barriers, language and cultural differences, and the anxiety of potentially losing access to promising therapies can discourage open conversations about adverse effects. Patients have specifically mentioned (see details below) that, sometimes, the very fear of side effects blocks treatment before it starts.
One of the most notable recent findings is how much patients depend on one another, and not just their healthcare providers, when dealing with treatment challenges. Genuine peer-to-peer conversations about tolerability offer essential support, counteract misinformation, and reduce feelings of isolation. Facilitating these community-based exchanges could play a crucial role in helping patients feel more empowered and knowledgeable when making difficult treatment decisions.
Beyond the Numbers: Toward Multidimensional Measurement
Traditional assessment tools tend to focus on the “what, when, and how severe” aspects of side effects. But capturing the full patient experience in clinical research or clinical care requires a broader lens that combines quantitative metrics (frequency, duration, severity) with qualitative insights: How does a side effect change a parent’s ability to work, or a child’s experience at school? Does this impact fluctuate depending on family dynamics or social support?
Disease-agnostic questionnaires and open-text responses are emerging as promising ways to surface the subtleties of tolerability, especially the individual stories that get lost in population-level data. Patients, caregivers, and clinicians seem to unanimously agree that incorporating both patient- and clinician-reported adverse events is essential for a more nuanced, patient-centric benefit-risk assessment.
Innovations in Patient and Caregiver Feedback
These novel approaches include a disease-agnostic qualitative tool for assessing patient perceptions of treatment benefits and side effects: the Patient’s Qualitative Assessment of Treatment version 2 (PQATv2). This 2020 measure, distributed by the Mapi Research Trust, has begun to be implemented in early-phase oncology studies. Preliminary use in phase 1 and phase 1/2 trials suggests that PQATv2 can enrich traditional efficacy and safety endpoints by capturing salient patient-reported concepts that may inform future PRO strategy. Early findings indicate it has value as an exploratory tool for generating hypotheses and guiding the selection of fit-for-purpose PRO measures in later development phases.
Patients, caregivers, and clinicians all see caregiver perspectives as a missing piece of the puzzle. Exploring ways to formally integrate caregivers’ observations, especially for patients such as children or those with cognitive challenges, could add a layer of insight to inform clinical research and care, as well as regulatory decisions. Capturing and analyzing open-text data from PROs such as the Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE®) also remains a challenge, with ongoing efforts in data science aimed at unlocking richer, more actionable insights from qualitative patient feedback.
Bridging the Communication Gap: Empowerment Through Dialogue
If measurement is one half of the tolerability equation, communication is the other. Patients and their families deserve to truly understand what to expect from a treatment, not just in terms of lab results or statistical risks, but also how side effects may alter their daily routines or long-term plans. Best communication practices to ensure this understanding include:
- Early, honest doctor-patient conversations about potential challenges with side effects
- Clear explanation of alternative therapies, including those with differing benefit-risk profiles
- Facilitating rapid connection to palliative care and supportive services when warranted.
Ultimately, equipping patients to make well-informed choices is more than a regulatory or scientific requirement; it’s a central pillar of optimal medicine and a driver of adherence, satisfaction, and real-world outcomes.
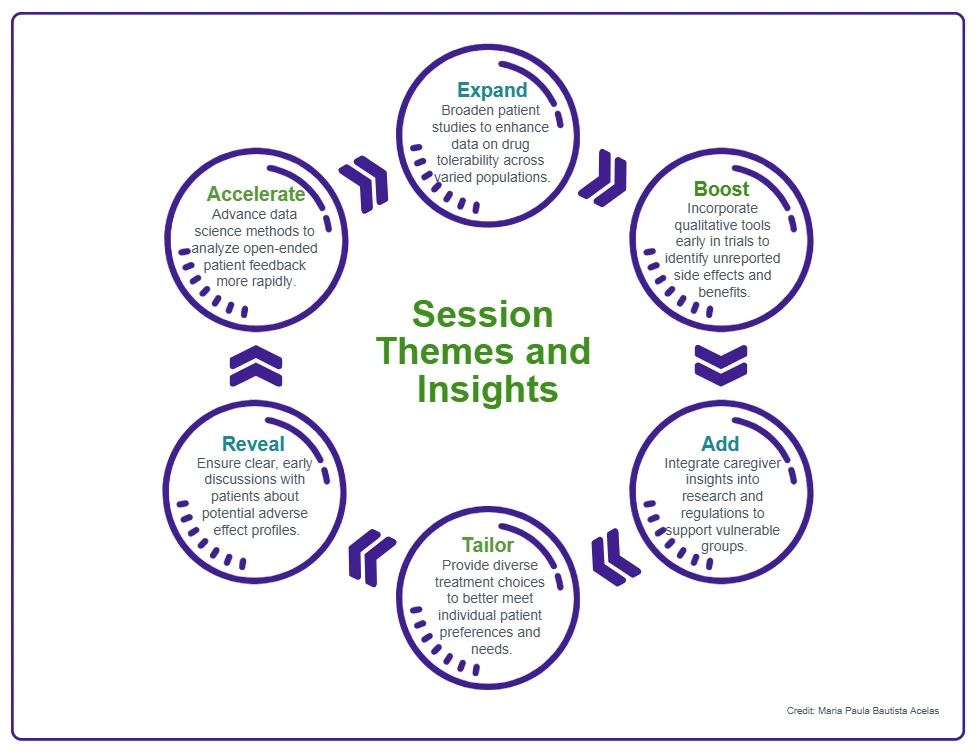
Session Themes and Insights
Themes and Insights in the infographic below build on the above and on other growing developments and expertise in this field:
Onward and Forward
The DIA Tolerability Study—a multidisciplinary collaborative working group focused on providing thought leadership in defining patient-reported tolerability, with an emphasis on immuno-oncology treatments for selected cancers and the goal of extending this definition to all cancer types—was launched in 2024. This study and related discussions (see below) have made the path forward clear: Incorporating patient stories and priorities is no longer optional; it’s essential for creating therapies that are truly effective and compassionate. By bringing the patient perspective into the definition of tolerability, we’ve only begun to reveal the depth and complexity of the issue. Significant work remains, but it cannot be done without continuing to listen, adapt, and innovate together. Then and only then will the future of drug development reflect what matters most: the diverse, meaningful lives patients want to lead. In the end, the evidence brings us back full circle: the patient knows best, and their perspective must remain the foundation upon which tolerability, as part of truly patient-centered drug development, is built.
DIA will continue to support this ongoing evidence-based initiative, which aims to build a foundation for understanding how cancer treatment tolerability is perceived worldwide. To learn more about this work, explore opportunities to contribute, or join a future work of the initiative, please contact science@DIAglobal.org.

