Tufts Center for the Study of Drug Development
early 40 years ago, the first regulatory guidance on monitoring clinical trials in the US was published by the US Food and Drug Administration (FDA). For the next two decades, traditional onsite monitoring was performed by Clinical Research Associates (CRAs)* who relied primarily on 100% Source Document Verification (SDV) as the predominant mechanism to fulfill regulatory requirements to monitor the progress of clinical trials and to ensure human subject protection and data integrity.
Ten to 15 years ago, a paradigm shift began to unfold in which regulatory agencies and Good Clinical Practice (GCP) guidelines started advocating for more risk-based, remote and centralized monitoring (CM) approaches. Industry has gradually embraced this approach as the FDA and the International Conference on Harmonization (ICH) among others have provided more clarity around risk-based quality management (RBQM) and risk-based monitoring (RBM) plans and approaches. Recent surveys providing benchmark data on the adoption rates of RBQM reinforce that this momentum is building.
RBM has the potential to have a significant impact on their responsibilities, perhaps more than any other role in the clinical operations ecosystem. CRAs play an essential role in the implementation of RBM. To date, however, data on CRAs’ experience with, and perception of, RBM has been limited to anecdotal reports suggesting mixed perceptions and reluctance to make the transition.
Key themes regarding CRAs’ positive and negative views of RBM include:
Positive perceptions:
- More efficient monitoring
- Greater focus on high-value activities and critical data
- Adaptive or triggered monitoring based on level of study and site risk
- More holistic view of study and site performance
Negative perceptions or concerns:
- More difficult to get site cooperation
- Overall skepticism about the negative impact to clinical trial oversight
- Concern over missed serious adverse events (SAEs) and protocol deviations
- Reduced site contact and negative impact on site relationships
- Insufficient training in the RBM process
- Increased workload and administrative burden
The lack of a systematic evaluation of CRA perspectives prompted a new survey conducted by the Tufts Center for the Study of Drug Development (Tufts CSDD) to assess the impact of RBM plans, particularly those including elements of CM, on field-based CRAs who conduct onsite monitoring visits. The survey targeted a subset of CRAs who could provide perspectives on how RBM approaches compared to traditional monitoring, including those who:
- Conduct onsite monitoring;
- Have monitored within the last two years or longer; and
- Have experience operating under RBM plans.
In-house study monitors who conduct remote, off-site, or centralized monitoring were excluded from participation in the survey.
Survey Overview
The Tufts CSDD research team developed two surveys following American Association for Public Opinion Research (AAPOR) guidelines: one targeting industry sponsors, and another aimed at CRAs, with input from a working group comprised of 16 major biopharmaceutical companies.
The results of the CRA survey are presented here; results from the sponsor survey will be published in a separate manuscript. The analysis included descriptive statistics, frequency comparisons, analysis of mean response values, subgroup stratification, and significance testing. Data cleaning and analysis was conducted by R (version 4.3.3), a robust statistical analysis program.
The CRA survey on clinical trial participants was distributed globally by Tufts CSDD and working group members using Qualtrics, an online survey platform, between May and July of 2025. Broadly, the survey aimed to gather CRA perspectives, based on their 2024 experiences, on the impact of RBM on monitoring workload, effectiveness, and site relationship quality. The survey captured data on different types of monitoring approaches, site visit schedules, and factors that trigger an onsite monitoring visit. The survey also explored different workload scenarios, the CRA’s ability to comply with adaptive site visit schedules, and experience with CM reports. The survey also gathered feedback on the impact of RBM on a variety of parameters, from data quality and protocol compliance to monitoring visit effectiveness and site relationships.
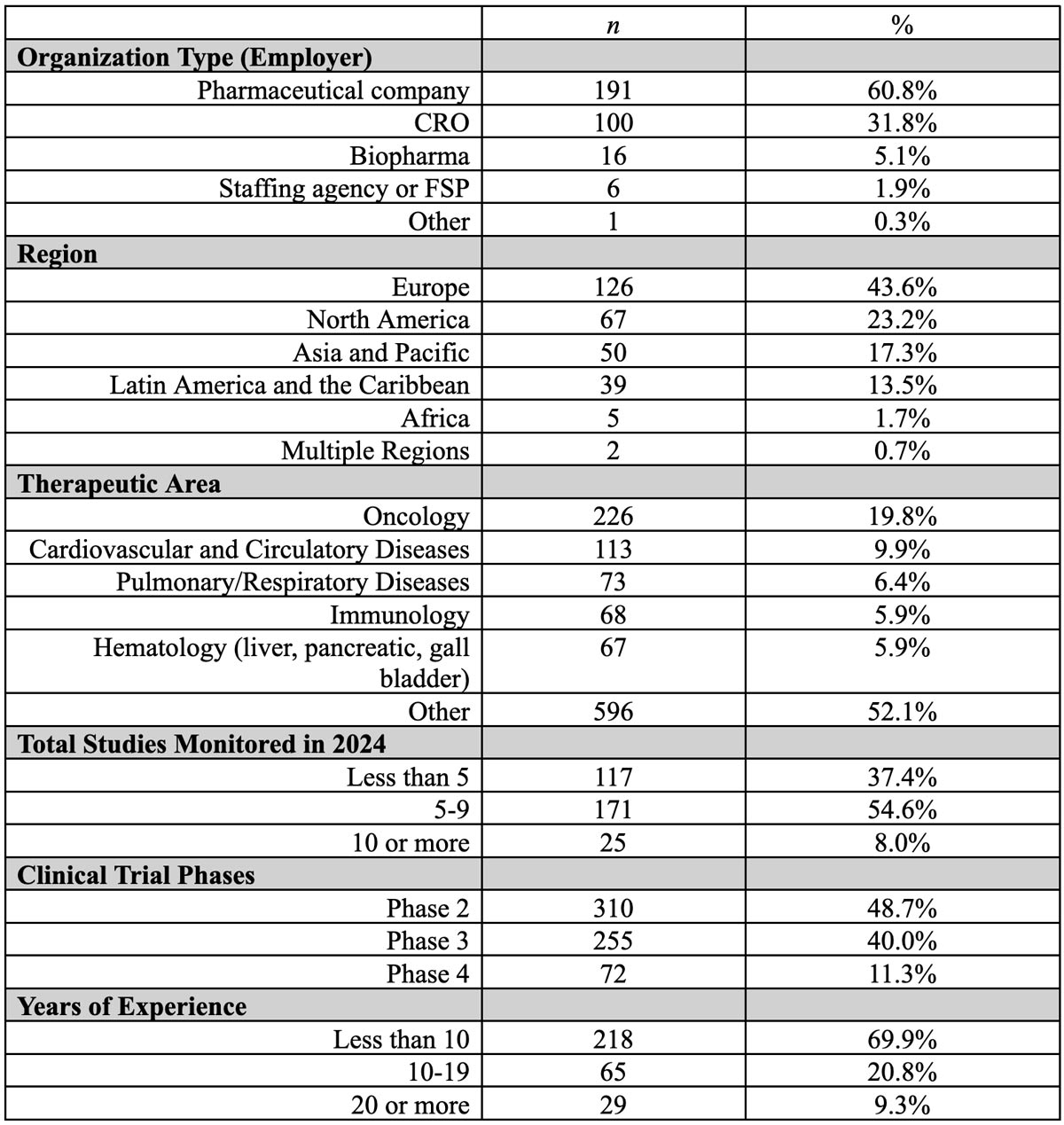
The survey received approval from the Institutional Review Board at Tufts University, and all respondents provided electronic consent to participate at the beginning of the survey. Table 1 presents the demographic characteristics of survey respondents, demonstrating broad representation across diverse groups.
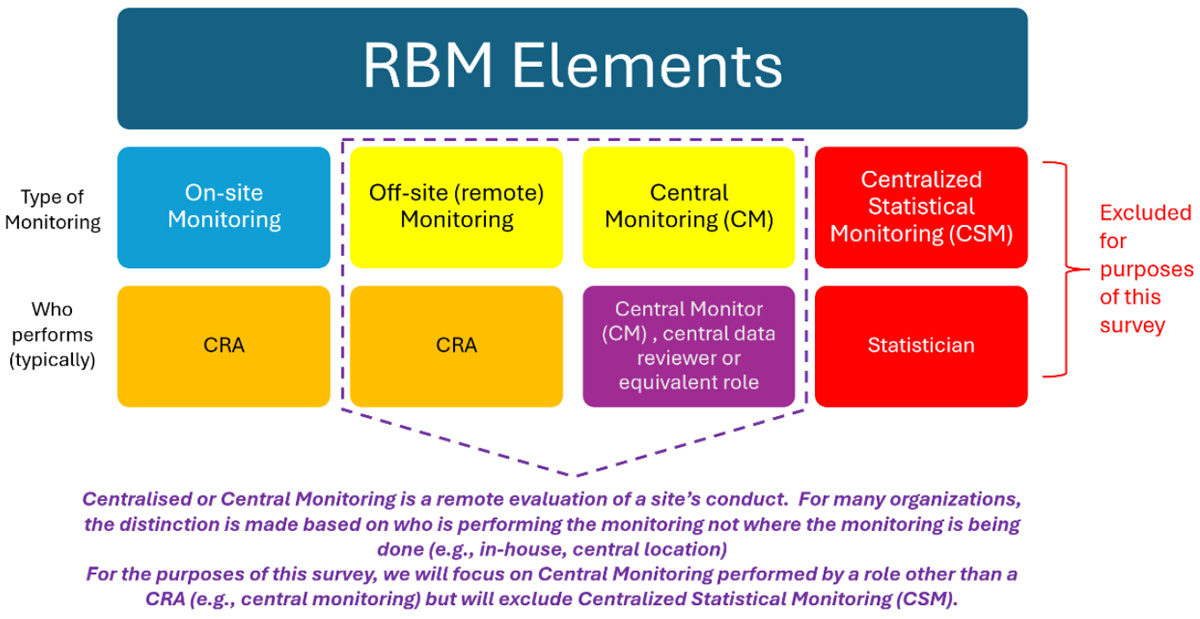
Definitions and Frameworks
To ensure a common understanding of terminology, definitions of different elements of RBQM/RBM were included in the survey. These represent a blend of various industry and regulatory definitions (Table 2). Recognizing that RBM is a subset of RBQM, we will refer primarily to RBM throughout this manuscript.


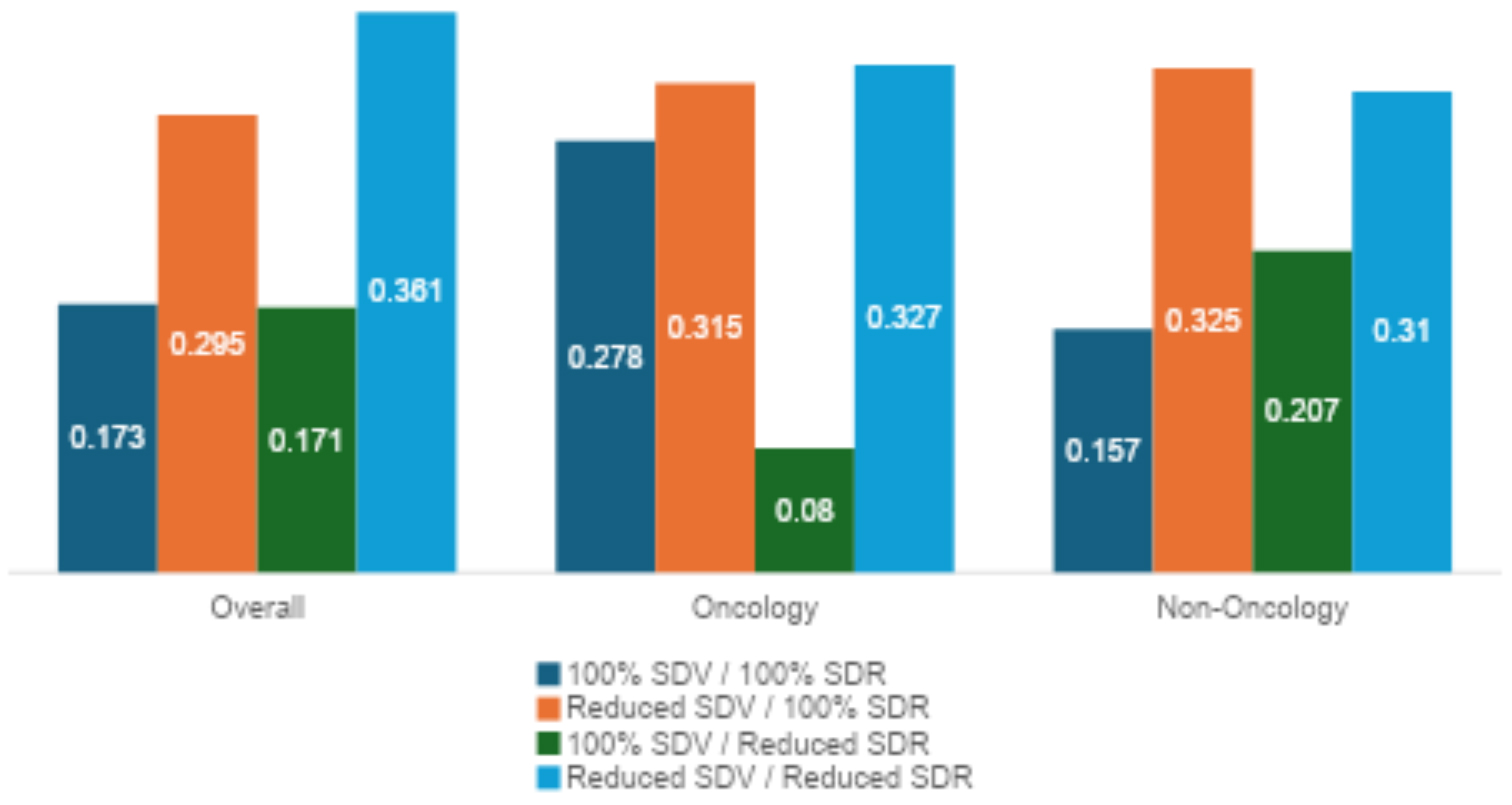
Changing Study Monitoring Approaches
The results of this survey revealed that adaptive data monitoring approaches were used widely in 2024 (Figure 3). CRAs reported that the most often used approach included combined reduced SDV and reduced SDR. Nearly three-quarters of the survey respondents report using a model combining reduced SDV but 100% SDR. The two other models (100% SDV + 100% SDR and 100% SDV + reduced SDR) were also used but at a reduced level compared to the other models.
There were some notable differences between oncology and non-oncology studies in terms of the different approaches used, with oncology studies using the traditional 100% SDV + 100% SDR model over 27.8% of the time compared to about 15.7% of the time for non-oncology studies. In addition, oncology studies used the 100% SDV + reduced SDR model about half as often as non-oncology studies.
About half (47.2%) of the respondents reported a reduction in the number of onsite visits since implementing RBM approaches. Only 14.5% of the CRAs reported that their onsite visits had increased as a result of the adoption of RBM, while 38.3% reported that the number of onsite visits had not changed.
In addition to describing the impact of RBM on the number of site visits, CRAs were asked to report on site visit frequency and duration as well. In a traditional monitoring approach, CRAs report that a typical schedule involves a site visit every 7 weeks, with an average of 2 days onsite and each visit comprised of 8 hours spent per day onsite (Figure 4).
For studies using an adaptive site visit schedule, the results show that the time between visits increases as the site risk level decreases; each sponsor independently defined “High,” “Medium,” and “Low” risk levels based on key study-specific risk indicators. High-risk sites are seen every 6 weeks on average compared to every 8 and 12 weeks for medium and low-risk sites respectively. CRAs reported spending an average of 1 day onsite for low-risk sites versus an average of 2 days/site visit for other site risk levels. The average time onsite per visit does not vary based on site risk and remains consistent with traditional and monitoring visit schedule at 8 hours onsite, with slightly less time (7 hours) onsite for low-risk sites.

- Site was not entering data and/or answering queries within the expected timelines;
- Site had a higher number of queries than other sites; and
- There was a data cut or interim analysis.
The next most common reason triggering a site visit was enrollment- or retention-related reasons (29.9%) and included:
- Site was not enrolling at the expected rate
- Site overenrolled participants compared to their target number
- Site enrolled at a faster rate than other sites
- Site had a higher number of screen-failed participants
- Site had a high number of early terminations.
The remaining triggers were requests from management, site, or the CRA for additional time onsite (17.7%). Interestingly, GCP/protocol compliance (13.6%) or participant safety-related (7.5%) reasons were rarely the cause for a site visit.
CRA Workload Considerations
The survey explored a variety of workload-related topics. On average, CRAs reported that they typically monitor eight sites in a phase 2 or 3 study under a traditional model. This did not change as a result of the RBM model: CRAs report they are still assigned an average of eight sites per study.
According to this survey, most CRAs (59.7%) report having sufficient time to complete their onsite monitoring activities. Nearly a quarter (26.0%), however, indicate they are not allocated sufficient time to complete their required tasks at the site. A small percentage (14.3%) of the CRAs reported having more than adequate time to complete their work during an onsite monitoring visit.
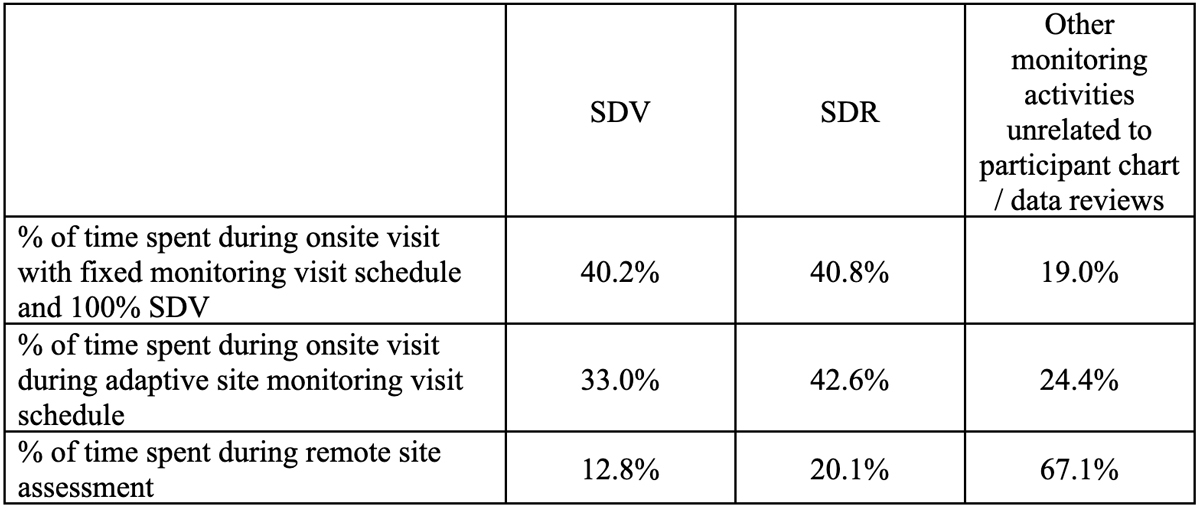
In terms of the distribution of activities performed by the CRA, they report a slight reduction in the time allocated to SDV during adaptive and remote visits compared to onsite visits (Table 4). The time allocated to SDR does not appear to vary based on the different monitoring approaches. Predictably, CRAs note a significantly greater percentage of their time is allocated to other monitoring tasks (outside of participant chart/data reviews) during remote visits.
CRA Views of Central Monitoring
CRAs generally find central monitoring (CM) reports helpful in terms of enabling or focusing their monitoring activities. The majority (62.9%) of respondents indicated that the reports are somewhat or very helpful, with only 15.6% indicating that they detract from more important monitoring activities. The remaining 21.5% of the CRAs indicated that the CM reports have no impact on their monitoring activities. Statistically significant differences (p-value <.05) were observed by therapeutic area and trial volume:
- A higher percentage of CRAs monitoring oncology studies—compared to those monitoring studies in other TAs—perceive the CM reports as somewhat or very helpful to enabling or focusing their efforts on more important monitoring activities.
- A higher percentage of CRAs monitoring 10 or more studies in 2024—compared to those monitoring a lower volume of studies—perceive the CM reports as less helpful (or more challenging).
Respondents provided an exhaustive list of comments, both positive and negative, pertaining to CM reports. From the need for better CRA and Central Monitor collaboration, to training needs, timing of the data reviews, and the content and format of the CM reports themselves, respondents identified many opportunities for improvement. Specifically:
- They call for much stronger collaboration and communication between the CM and the CRA in order to discuss the nuances of findings in CM reports and ensure that CMs have a more holistic view of the site’s performance.
- CRAs also advocate for more concise reports and clearer, simpler, and less ambiguous dashboard presentations of the data.
- They also desire more insights about what the data is conveying so that the information is more actionable in terms of where and how the CRA should focus their monitoring efforts.
- Additionally, many CRAs expressed frustration with the timeliness of the reports, noting that many are delayed or arrive late resulting in more rework to determine what information is outdated.
- Finally, CRAs expressed the need for better training of the Central Monitors, specifically around the protocol, indication, and the role of the CRA, that goes far beyond just checking source data.
CRA Views on the Impact of RBM
CRA views regarding the impact of RBM on various factors varied widely. CRAs report that the greatest improvement from RBM is on monitoring efficiency, effectiveness, and workload (Table 5). Over 60% of the respondents indicated that these areas have improved somewhat or greatly as a result of RBM approaches. In contrast, over one-third of CRAs perceived that RBM has had a negative impact on data integrity (41.5%) and data quality (36.6%). Views on the impact on other operational considerations, such as participant safety risks, the strength of site relationships, protocol compliance, and subject enrollment, are mixed.
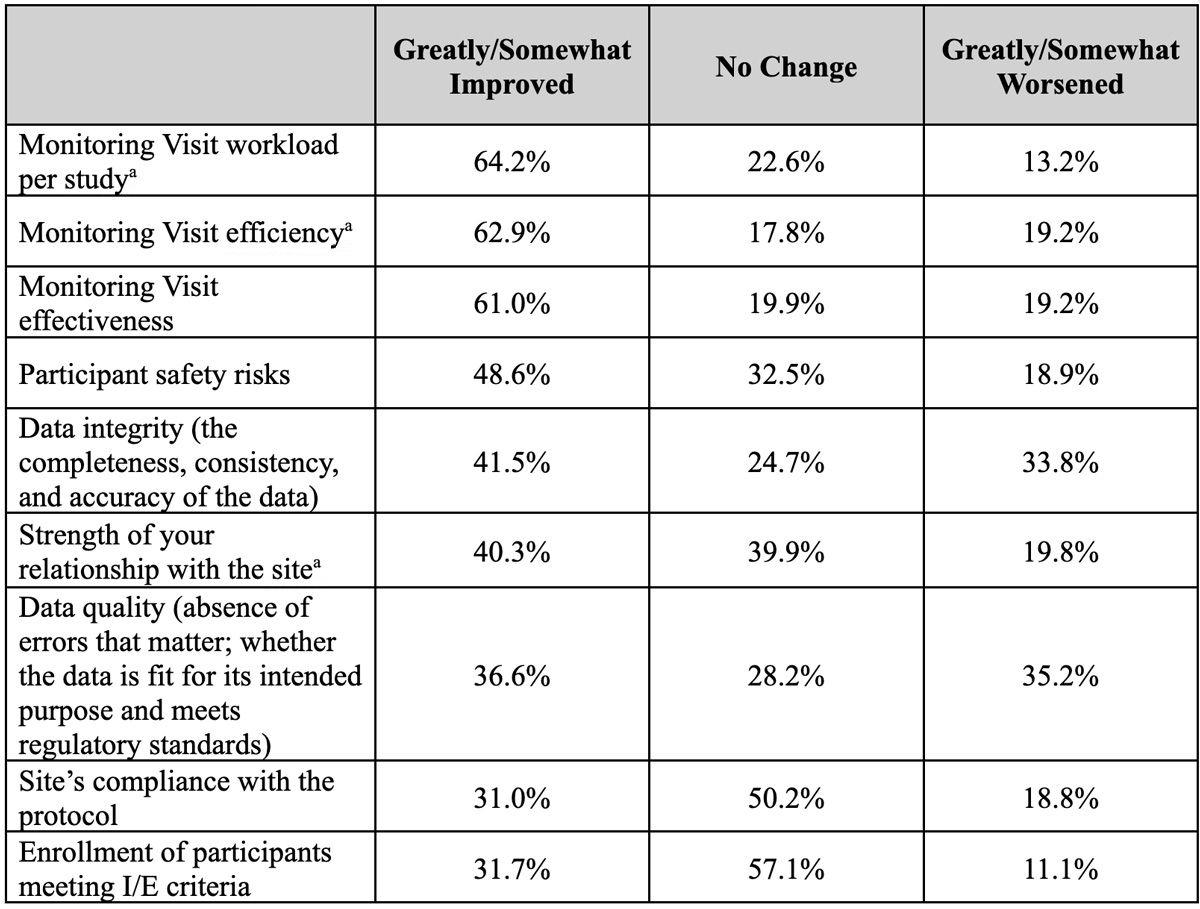
aA statistically significant difference was observed between CRAs monitoring oncology vs. non-oncology studies, with fewer CRAs in oncology studies noting that these areas had greatly worsened (p-value <.05).
CRAs report high compliance with adaptive visit schedules, which may contradict some anecdotal perceptions that CRAs are resistant to RBM approaches. Most CRAs (89.1%) report that they have been able to fulfill the RBM approaches that incorporate a scheduled adaptive site visit. Statistically significant differences were observed by therapeutic area as well as by years of monitoring experience. CRAs monitoring oncology studies perceived having somewhat or much greater ability to comply with the monitoring plan compared to their counterparts monitoring non-oncology studies. CRAs with 20+ years of experience reported having somewhat or much lower ability to comply with the monitoring plan compared to CRAs with less than 10 years of experience (p-value <.05).
Of the 10.9% reporting an inability to comply with the adaptive site visit schedule, key reasons included discomfort with the plan, site requests for more frequent onsite visits, or several human resource-related (HR) factors. With the low incidence of sites requesting more frequent onsite visits, this suggests they are also comfortable (or gaining comfort) with the approach, although this is an area requiring further investigation.
CRA Preferences and Recommendations
CRAs offered extensive feedback related to implementation of RBM from a qualitative perspective. Most comments (46.7%) related to the monitoring plan itself, with a mix of those in favor and noting the need for adjustments to those who believe RBM is not adding value. Technology-related observations were the next highest category (20.5%) where CRAs offered suggestions. All other topic areas were noted by less than 10% of the respondents and included recommendations related to workload, resources, training, and site relationships.
CRAs who oppose RBM primarily reinforce many of the same concerns noted above:
- Increased workload from cross-referencing multiple reports or chasing invalid issues / requests
- Site resistance to remote monitoring visits that are deemed “unproductive, redundant and time consuming for site staff”
- CM reports that are not tailored to the specific site or participant.
Despite the perception by some CRAs that remote monitoring visits may not be productive, the majority of respondents indicated they would actually prefer more remote visits (or somewhat less time on site) than their current model (Figure 4). They describe an approximate 10% shift in their ideal mix of onsite versus remote monitoring compared to the actual distribution they experienced in 2024.

Other CRAs, however, are strong champions for RBM. Similarly, they reinforce the positive aspects previously noted, including optimizing activities that provide higher value and allowing more time for site relationship management. On the other hand, they caution sponsors to be realistic about the financial and efficiency gains professed by RBM, which may be offset by increasingly complicated protocols that necessitate more work by the CRA.
As noted above, technology factors received the second highest number of comments. Topics included artificial intelligence and CM reports, EDC/EMR and other system integration considerations, as well as the need for standards and best practices surrounding technology platforms. Overarching themes and recommendations centered around the need for:
- Better EDC systems that make it easy to identify which data points require SDV
- Improving data-handling tools
- Better cross-system integration into a one-stop shop for CRAs to increase their confidence and efficiency to identify relevant information
- Developing data systems with the assistance of experienced end users who can provide valuable insights into what is and isn’t working.
RBM Transforming Traditional Roles for CRAs
While not yet fully mainstream, RBM adoption is increasing. More and more CRAs are gaining familiarity with adaptive monitoring visit schedules, reduced SDV/SDR approaches, and use of CM reports. They are experiencing the impact that RBM overall, and different site-risk levels specifically, are having on their workload. They are learning (or in some cases relearning) how to prioritize tasks during onsite monitoring visits, and they are adjusting to a different mix of remote versus onsite visits to accomplish their work.
In essence, the traditional role of the CRA is undergoing a significant transformation as a result of RBM. Inevitably, such a dramatic shift in responsibilities would cause consternation, particularly for seasoned CRAs with years of experience operating under the traditional monitoring model. To date, little has been known about how CRAs view the transition to RBM beyond anecdotal reports.
This study provides the first-ever comprehensive understanding of CRAs’ experience with, and perceptions of, RBM. The results corroborate other benchmark data that indicates a growing number of organizations are implementing RBM approaches. Further, the results indicate that RBM is surpassing the traditional high site visit frequency with a 100% SDV model. While a fixed-visit schedule is still widely used, other adaptive visit schedules are being implemented nearly at the same rate. Generally, CRAs have seen a reduction in the number of onsite visits with RBM and note about a quarter of their work is completed remotely. While the overall number of sites allocated to a CRA on average hasn’t fundamentally changed, the categorization of sites based on different risk levels has had significant bearing on site visit frequency. While the study did not define the site risk level categories explicitly, respondents reported that high-risk sites are seen twice as often as sites deemed to be low risk. From a data monitoring perspective, reduced SDV/SDR models are now more common than the traditional 100% SDV + 100% SDR approach of the past.
Overall, CRAs report that RBM has greatly helped them focus on more high-value or high-impact areas during onsite monitoring visits. They describe little difficulty complying with the adaptive site visit schedules. CRAs generally find CM reports helpful in terms of enabling or focusing their monitoring activities, but see substantial opportunities to improve the content, format, and timing of these reports.
CRAs report that monitoring workload, efficiency, and effectiveness have improved as a result of RBM; however, they still have concerns about RBM’s negative impact on data integrity and quality. Opinions vary as to RBM’s impact on site relationships, participant safety, protocol compliance, and subject enrollment, suggesting that positive impact in these areas has yet to be realized.
RBM is Here to Stay
Industry has moved beyond questioning the rationale and regulatory requirements for RBM to implement adaptive monitoring methods and support the use of CM technologies. While some hesitancy remains, this study suggests that CRAs are adapting to, and in some cases embracing, their transformed role and have rich experiences to share. By soliciting insights and feedback from CRAs, Sponsors, CROs, and other service providers have an opportunity to refine their RBM methodologies and CM technologies. In doing so, CRAs may evolve from reluctant participants to full-blown advocates of an approach that enables them to focus on the highest value activities and what really matters in the conduct and oversight of a clinical trial.
Future research will explore CRA line manager views on RBM to better understand the impact that adaptive site visits and CM have on CRA training, workload allocation, and performance evaluation. Evaluating the impact of RBM on investigative sites is also warranted so that their needs and experiences can be accounted for to ensure the effective implementation of these adaptive monitoring approaches.
Acknowledgements
The authors thank all the 16 sponsor companies that participated in the study’s working group and the individual representatives who contributed their ideas and expertise to the design of the survey. The authors also thank the field CRAs who responded to the survey and provided their insights and thoughtful recommendations.
Additional references available upon request.

