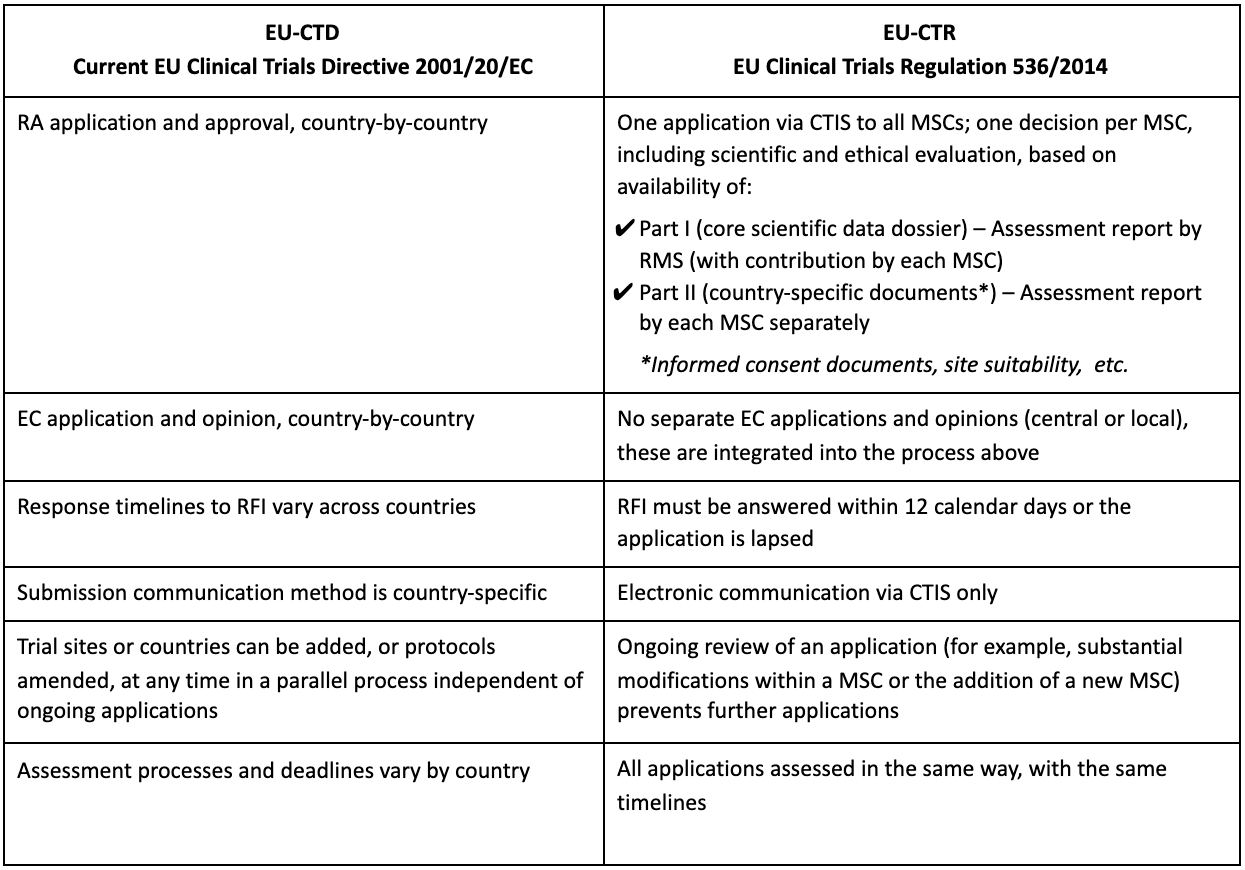
he European Union Clinical Trial Regulation 536/2014 (EU-CTR) aims to standardize and harmonize the conduct and management of interventional clinical trials across the European Economic Area (EEA), with legally binding rules on requirements and increased transparency.
- submit, evaluate (via scientific and ethical review), and authorize clinical trial applications (CTAs);
- submit any trial-related notifications, reports, and results, up to the clinical study report; and
- serve as the single communication channel between the sponsor and the Member States Concerned (MSC) in the clinical trial.
December 2021 is only one year away, and preparing for the EU-CTR requires wide-ranging, cross-company initiatives. Companies should mobilize all stakeholders to analyze current business processes, upgrade information technology systems, and restructure operations to avoid disruptions to start-up, conduct, and maintenance of ongoing and new trials.
To Reap Efficiencies, Sponsors Must Sow Operational Changes
Efficiencies promised by the EU-CTR include all-electronic submissions and communication, improved collaboration between Member States (MS), increased process transparency, and uniform review timelines. However, EU-CTR presents significant operational challenges for sponsors. Although core activities and datasets for trials in the EEA will not change, management of study start-up activities, RFI responses, and modifications to the CTA dossier need to change. For example, sponsors must establish processes for document redaction.
Here are essential preparations companies should undertake now:
- Analyze processes across your portfolio
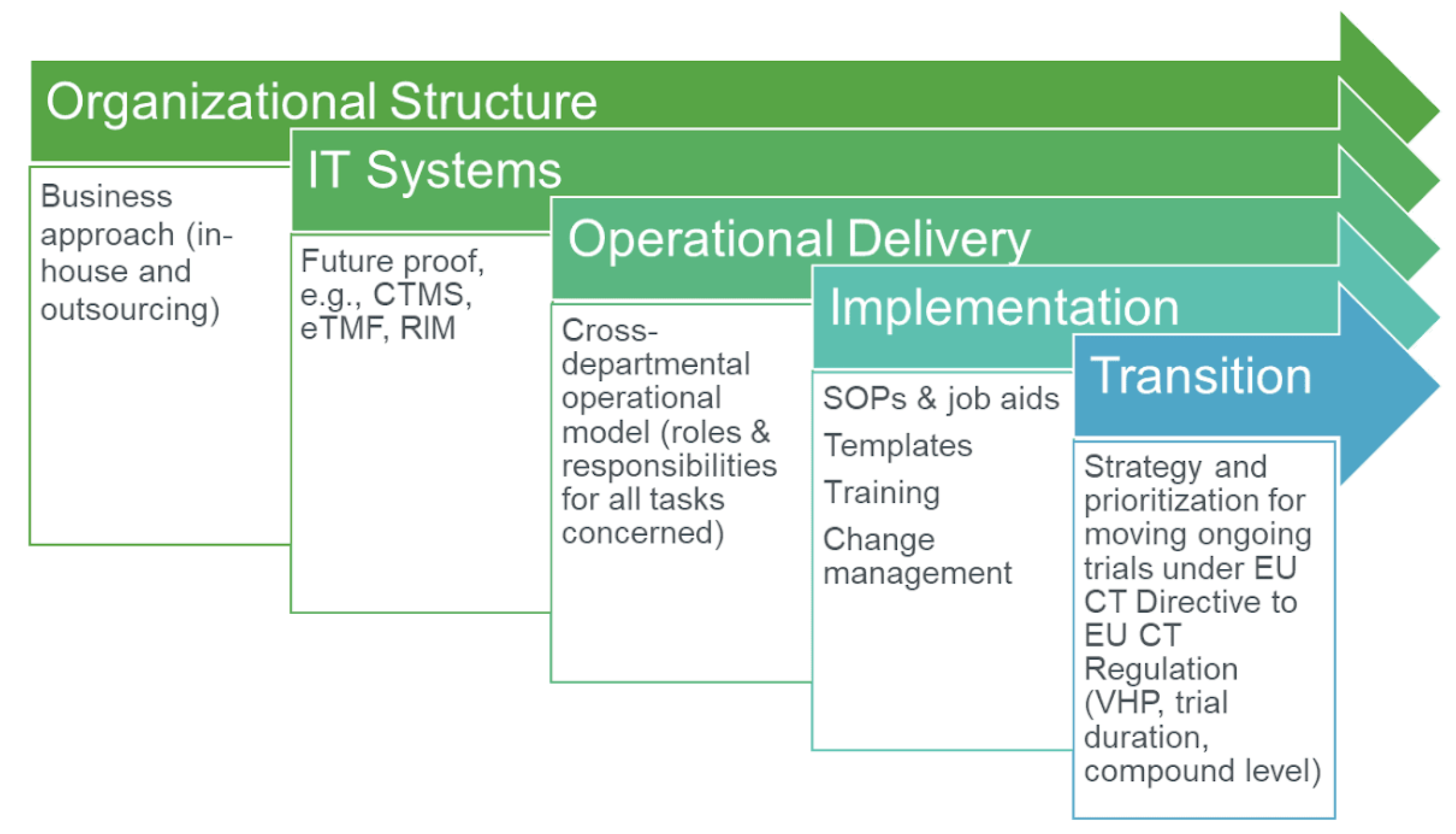
To maximize efficiency, sponsors need to assess processes across their portfolio lifecycle. Early on, sponsors should engage with internal stakeholders and external outsourcing partners to communicate clearly defined processes and workflows to ensure business continuity and full compliance under the EU-CTR. They may need to employ different resourcing models to address varying circumstances.Given the time to implement all required changes, sponsors must prepare for an EU-CTR future before they know all the pertinent details and may have to consider multiple contingencies (Figure 1).
- Focus on first-time quality
Under the EU-CTR, a CTA’s success depends on the first-time quality of the dossier to a greater extent than ever before. A CTA will not pass the validation phase in any MSC unless it receives clearance to proceed in all MSCs. The initial application authorization procedure and any Substantial Modification application must be completed before you can submit an additional MSC. This greater interdependency amongst MSCs in the trial demands that sponsors get applications right the first time. - Choose countries wisely
Under the EU-CTR, the study start-up strategy will need to change from “first EEA country ready” to a holistic approach to EEA country readiness. Part I of an EU-CTR CTA dossier evaluation is a joint assessment from all MSCs led by the RMS (sponsors propose the RMS). However, if a CTA contains elements or provisions at odds with the RMS’s national laws, a negative Part I conclusion will affect all MSCs. Experts with local knowledge can tailor the country list; choosing the right RMS and MSC will be critical. - Centralize CTA management
An industry-wide trend related to EU-CTR is the centralization of CTIS management. Communication exclusively via CTIS and the short RFI response timelines call for a focused team that monitors CTIS for incoming communications, handles document/data entry and download for trial master file compliance, and closely monitors timelines. - Upgrade IT infrastructure
Most companies will need to upgrade their electronic trial master file, regulatory information management, and clinical trial management systems to meet the new demands of EU-CTR. However, companies should realize that procuring and upgrading such systems can take anywhere from six to 12 months. And you can only upgrade effectively after a comprehensive impact assessment. It is vital to get the right insight and information about the current IT system and what will be needed going forward. - Tighten timing
Under the EU-CTR, sponsors will need to coordinate initial applications—and all modifications—closely. Missed timelines may result in legal consequences for sponsors, such as lapsed applications. Sponsors will need a strategic plan for management and oversight of timing of permitted and non-permitted overlapping initial, substantial modification, and additional MSC applications.EU-CTR will require tight process coordination by sponsors’ study teams: all contributors must synchronize activities, both lifecycle planning for each trial as well as across all studies with the same product, because an ongoing assessment in one MSC will block further substantial modifications to Part I, or Part I and II, for all MSCs. Sponsors should attempt to minimize in-process changes to a clinical trial and ask: Will this action impact the submission of another action?
If sponsors prepare well in advance, they will find that initiating and conducting clinical trials in a timely fashion will be manageable under the EU-CTR.

