Uppsala Monitoring Centre
Decision Analytics LLC
Johns Hopkins University School of Medicine, Baltimore, MD
Boehringer Ingelheim
he Identification of Medicinal Products (IDMP) standards establish a global framework for uniquely describing medicinal products consistently across jurisdictions. When widely and consistently used, IDMP enables reliable cross-border product identification, faster recall and shortage responses, improved pharmacovigilance, and efficient regulatory reliance.
Moving from Dialogue to Action
A DIAmond session convened during the DIA Global Annual Meeting 2025 assembled regulatory authorities, industry leaders, global health organizations, and standards bodies to review the state of IDMP implementation and its relevance to global supply resilience. Following this DIAmond session, a Solution Room was convened to focus on practical, collaborative problem-solving. This article synthesizes the key insights from those discussions, highlighting barriers, root causes, and actionable strategies across four critical focus areas: Regulatory Alignment, Data Standardization, Industry Adoption, and Patient-Centric Benefits.
Regulatory Alignment and Global Harmonization
The Challenge
Regulators are implementing IDMP on different timelines, at different levels of granularity, and with varying expectations for data submission and validation. This results in the same medicinal product being represented differently across regions, complicating therapeutic equivalence assessments and limiting the ability of regulators to rely on one another’s decisions.
Root Causes
These differences stem from the absence of a coordinated global governance structure and limited shared operational guidance. Some agencies have begun to integrate IDMP data into review workflows (see examples from ANVISA in Brazil, EMA, and the US FDA), but others are still assessing readiness, which results in uneven progress. Industry is often required to generate multiple variations of identity data to meet differing regional regulatory expectations, leading to duplication and slowed adoption (Table 1).
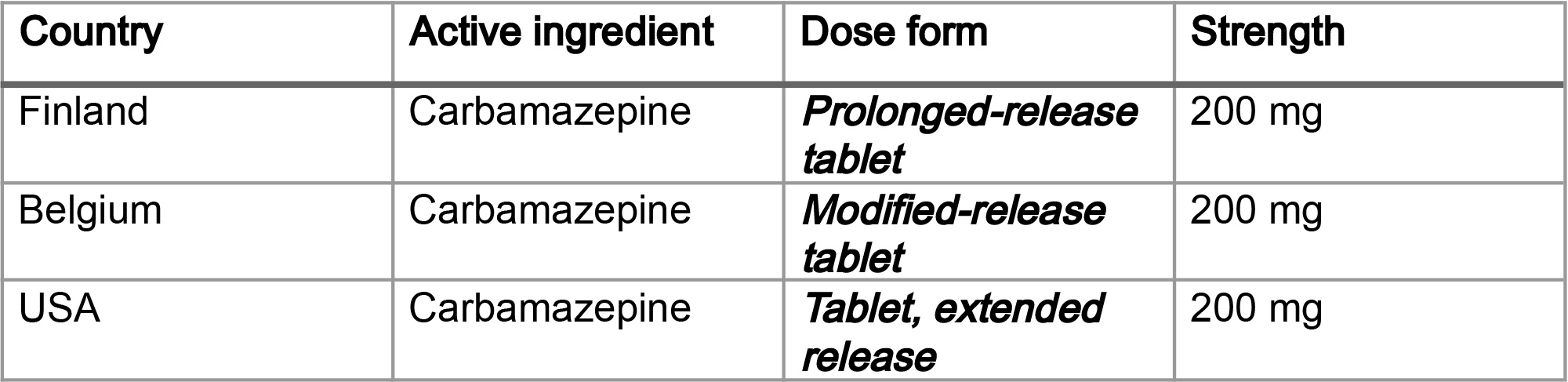
More consistent operational guidance and shared reference models that support both flexibility and convergence are needed. Coordinated technical assistance and capacity-building, especially for regulators working toward digital transformation in their own agencies, would reduce fragmentation and establish clearer expectations for industry.
Proposed Way Forward
A globally coordinated implementation roadmap was identified as a priority. This roadmap would not impose a single mandatory implementation model but would articulate shared milestones, core data expectations, and pathways for regulatory reliance and work-sharing. Similar initiatives have been started by ICMRA-ICH-IPRP-PICS to develop a joint work plan for harmonization and convergence work that advances development of a regulatory pharmaceutical quality knowledge management capability.
The proposed identifiers framework to enable Pharmaceutical Quality Knowledge Management (PQ KM) evaluated by ICMRA would be a useful resource for a globally coordinated implementation roadmap.
Data Standardization and Technology Integration
The Challenge
Medicinal product data resides across many regulatory, manufacturing, labeling, supply chain, and safety systems, and is rarely structured in a way that supports interoperability. Legacy terminologies and system-specific representations can lead to inconsistent identification and interpretation of similar products.
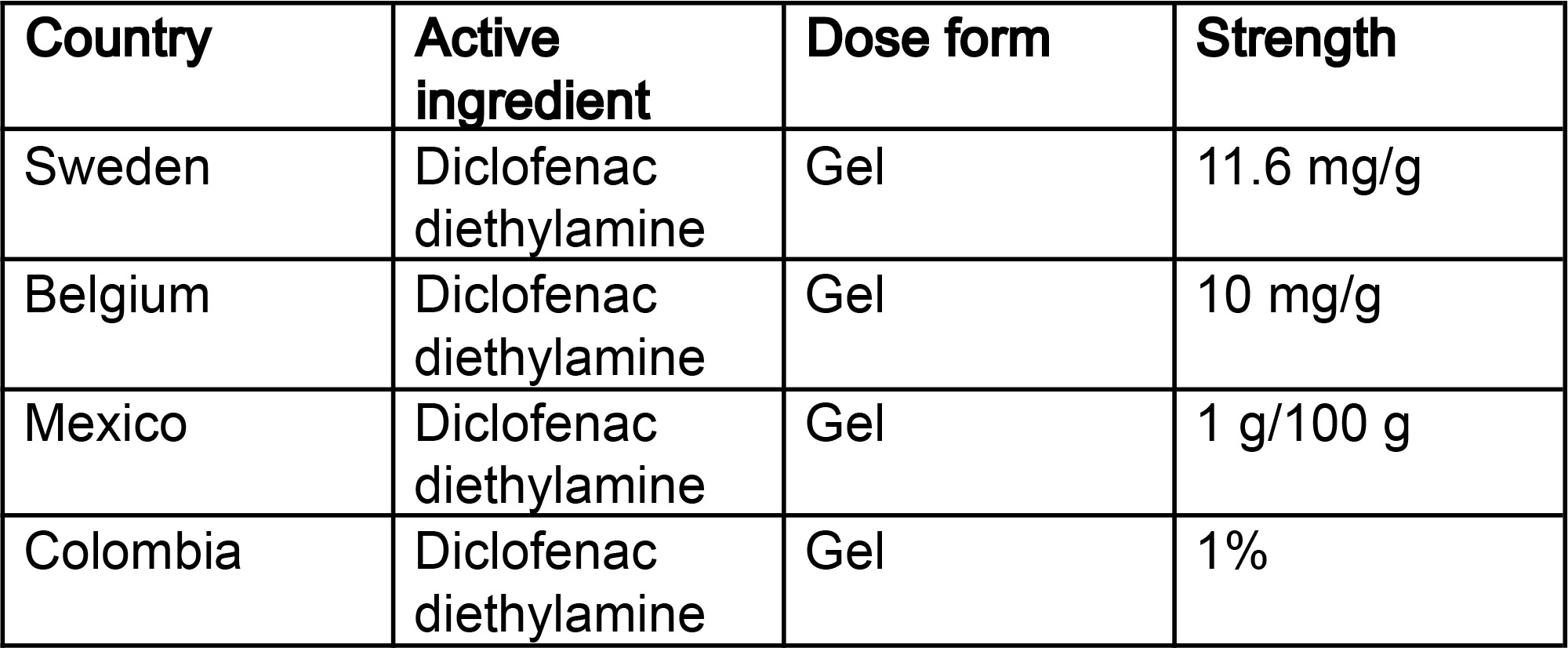
Countries often prioritize maintaining control over their own regulatory and product information systems, which can make alignment across borders challenging. In many cases, medicinal product data have been built up over decades within legacy systems that use local terminology, formats, and classifications. Converting these data sets into IDMP-aligned structures requires technical resources and deep data stewardship expertise. However, not all organizations have the capacity or familiarity needed to carry out this work at scale, resulting in slow and uneven progress. Consequently, legacy data persists and variation in how product information is represented continues to limit interoperability.
Suggested Solutions
Applying advanced technology to support data harmonization can address many of these challenges: AI-enabled tools trained on globally agreed-upon rule sets can accelerate mapping and reconciliation of legacy data. Knowledge graph–based approaches can help compare product attributes, clarify relationships, and identify differences in records across jurisdictions. Establishing single authoritative sources of product identity within organizations can reduce duplication and support consistent downstream data use. Additionally, embedding compliance capabilities directly into systems and workflows, instead of manually installing required updates, can help sustain alignment over time.
Proposed Way Forward
These experts recommended developing a common rule-based reference for guiding legacy data conversion, paired with machine-readable standards that can be interpreted and validated in automated systems. This approach would allow organizations to scale implementation more effectively, reduce manual effort, and maintain alignment as data evolves over the product lifecycle.
Industry Adoption and Implementation Hurdles
The Challenge
Industry participation is essential to realizing the full value of IDMP. However, in many organizations, these standards are still viewed primarily as a regulatory compliance obligation and not an enabler of operational efficiency and global product consistency.
Due to inconsistent implementation across companies and therapeutic areas, IDMP data is frequently created solely for regulatory reporting and then left unmanaged, failing to be maintained throughout the product lifecycle.
This limits the ability to use the data to streamline submissions, coordinate labeling changes, or support supply and safety decisions.
Root Causes
Several factors contribute to this dynamic. National Regulatory Authorities differ in infrastructure maturity, priorities, and statutory requirements, which can create unclear or inconsistent infrastructure expectations across regions. In addition, industry is not always positioned as a collaborative partner in shaping implementation models, which can reduce ownership and shared accountability. Finally, when IDMP is not integrated into routine lifecycle and quality processes, the underlying data quickly becomes outdated, which dilutes its value.
Suggested Solutions
Reframing IDMP as a foundation for global product sameness, regulatory reliance, and coordinated lifecycle management remains critically important. Aligning IDMP with complementary initiatives such as eLabeling and ePrescription systems can also help demonstrate practical relevance. Participants noted the benefit of creating structured opportunities for organizations to share implementation lessons, governance models, and demonstrated value cases. Embedding IDMP requirements into GxP frameworks and internal accreditation processes can further reinforce sustainability and lifecycle alignment.
Proposed Way Forward
Stronger, more proactive engagement pathways between industry and regulators will be necessary to advance shared implementation goals. Demonstrating the operational value of IDMP, from clinical development and manufacturing through patient administration, will help shift adoption from a compliance-driven exercise to a strategic enabler of efficiency, transparency, and patient safety.
Patient-Centric Benefits
The Challenge
Although the central purpose of IDMP is to support safer and more informed medication use, the benefits of its standards remain largely invisible to patients and healthcare providers. Limited integration of IDMP identifiers into electronic health records, pharmacy systems, and clinical decision support tools means that the standard has not yet translated into improvements in everyday care. As a result, patients are often unaware of IDMP’s role in ensuring product consistency and transparency across the supply chain.
Root Causes
One of the key reasons for this lack of visibility is that data standards work largely “behind the scenes,” which makes it difficult to communicate their value to patients and providers. Transitioning clinical systems to new data structures can also raise concerns about workflow disruption or delays in medication access. In addition, there are few if any clear, accessible educational materials that explain how standardized product identity supports safer prescribing, substitution, and recall communication.
Suggested Solutions
Addressing this gap requires elevating IDMP from a technical framework to a tool that directly supports patient-centered decision-making. Developing straightforward educational resources for clinicians and patients can help clarify why standardized product identities matter. Linking IDMP implementation to initiatives that have immediate public health relevance—such as adverse event reporting, drug shortage response, and recall coordination—can further highlight its value. There is also an opportunity for industry to develop patient-facing tools, such as a digital medication card or “wallet” that uses IDMP identifiers to provide accurate, portable medication information. This will make benefits tangible for clinicians and patients and create support for broader rollout. Collaboration with pharmacies and electronic health record vendors will be essential to integrate these capabilities into routine care.
Proposed Way Forward
The roundtable emphasized the need for sustained partnerships with patient organizations, pharmacists, and healthcare delivery networks to ensure that IDMP adoption advances in a way that strengthens care rather than complicates it. Demonstrating the benefits of standardized product identity at the point of care will be important to making IDMP meaningful and visible.
Key Takeaways
- Each stakeholder group has a distinct but complementary role in advancing IDMP.
- Regulators’ priority is to work toward greater harmonization of expectations and coordinated governance structures. Aligning timelines, data requirements, and implementation milestones will help reduce fragmentation and improve opportunities for reliance and work-sharing.
- Industry must shift its perspective from viewing IDMP as a reporting obligation to recognizing it as a foundation for lifecycle efficiency and global product consistency. Sustained value will come from embedding IDMP into routine operational and data governance processes rather than treating it as a one-time compliance project.
- Engagement and practical tools will be critical for patients and healthcare providers. Clear communication and user-friendly interfaces are needed to make the benefits of standardized product identity visible in clinical and pharmacy environments, where they can meaningfully improve medication safety and substitution decisions.
Concluding Call to Action
These conclusions reflect a shared commitment to shift IDMP from a regulatory requirement to a practical tool that strengthens the supply chain, enables coordinated regulatory decision making, and improves patient safety. Delivering on this commitment will require sustained collaboration among regulators, industry, standards organizations, and healthcare partners, supported by clear governance to set priorities and sustain momentum.
As the pharmaceutical ecosystem evolves, IDMP offers a rare opportunity to establish a single global language for medicinal products. This shared foundation will increase transparency, improve consistency, and support better informed decisions across the product lifecycle. By following the approaches outlined above, stakeholders can accelerate adoption, reduce unnecessary complexity, and advance IDMP’s central goal: better information that leads to better health outcomes.

