Around the Globe
rom September 30 through October 4, the African Union Development Agency (AUDA-NEPAD) and the World Health Organization (WHO), with support from multiple partners, organized a gathering in Victoria Falls, Zimbabwe. The week started with a scientific conference at which regulators, academics, industry, and African and global partners discussed the progress and lessons learned from implementation of regulatory harmonization in Africa, followed by a regulators-only meeting.
Top Organic Chemist Challenges Regulators: Create Transparent, Predictable Ecosystem
In his keynote address, charismatic Cambridge-trained synthetic organic chemist Professor Kelly Chibale from the University of Cape Town underscored the critical need for Africa to create an attractive environment for clinical research for R&D companies and organizations to undertake clinical trials in Africa, as well as enable African talent to stay in Africa and grow its own native R&D and clinical research sector. Kelly argued that if navigating regulatory approvals is made fully transparent and predictable in Africa, that attractiveness will create job opportunities for African scientists and enable development of drugs which take into account the unique genetics and socio-economic context of patients in Africa.
African Medicines Agency (AMA)
The African heads of state and government endorsed AMA in February 2019. The AMA treaty is in the process of national ratification – 15 parliamentary ratifications are required for AMA to become an institution of the African Union. It is expected that this ratification process will take two to three years based on experiences with other African Union treaties.
Between July and October 2019, the Boston Consulting Group, commissioned by the AU Commission and the Africa Union Development Agency (AUDA NEPAD), developed an AMA Strategy and Business Plan. The strategy and business plan were approved by the heads of regulatory agencies from across Africa meeting at Victoria Falls. Below we highlight three elements from this Strategy: the vision, the proposed value proposition, and what AMA will focus on in its first five years of existence.
High-Level Vision of AMA

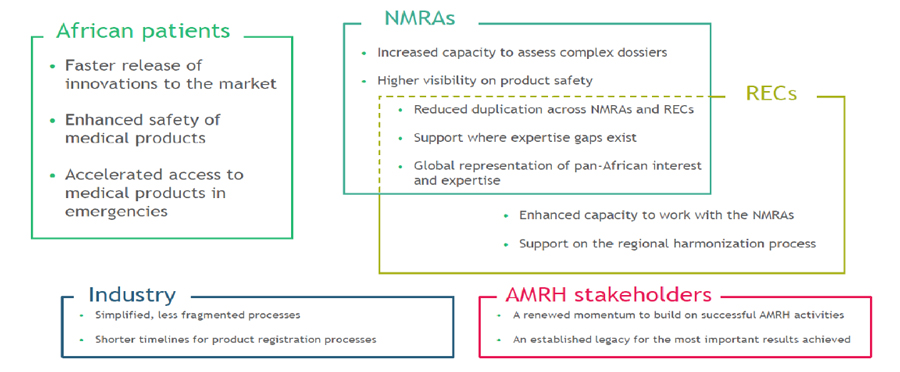
Proposed Value of AMA to Stakeholders
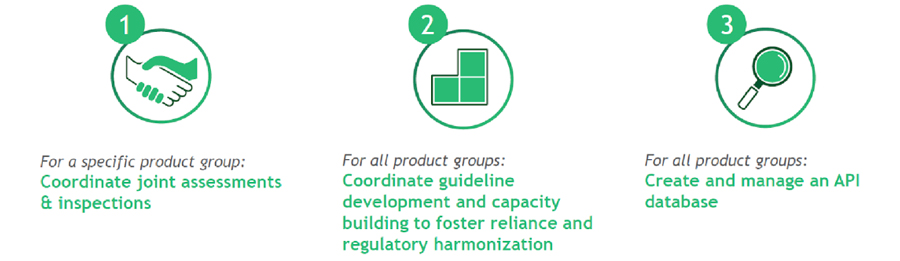
Three Things on Which AMA Will Focus in the First Five Years
What Will Follow?


