Graduate School of Medical Science, Kyoto Prefectural University of Medicine
apan formulated the “Third Term of the Health and Medical Strategy” (hereinafter referred to as the “Third Strategy”), with the approval by the Cabinet in February 2025 based on Article 17 of the Act on Promotion of Healthcare Policy. This overall Strategy aims to consolidate the government’s measures for research and development in the medical field. The Strategy considers current situations such as the decline in Japan’s research capacity and the issues of drug lag and drug loss, and outlines the future direction of research and development, basic policies, and specific measures for the five years from fiscal year 2025 to fiscal year 2029.
One major change from the Second Phase of the Strategy, which identified specific therapeutic areas that impact healthy life expectancy for further R&D and emphasized the importance of promoting healthcare services provided by private (non-public insurance) companies, is the addition of two integrated projects: Infectious Diseases, reflecting lessons learned from the pandemic; and Translational and Clinical Acceleration, emphasizing the importance of clinical trials. This article overviews the Third Strategy and introduces the progress made to date, focusing on new measures and those related to infectious diseases and clinical trials and clinical research.
Ambitious Output Indicators
The Third Strategy sets more ambitious output indicators (see Table 1) than the previous versions. These include initiating development for 86 pharmaceutical products currently affected by drug loss (see slide 12 of PMDA presentation), increasing target numbers for development plans and designations of pediatric medicines and orphan drugs for rare diseases, and expanding investment in drug discovery start ups. In particular, the goal is to significantly expand clinical trials both quantitatively and qualitatively.
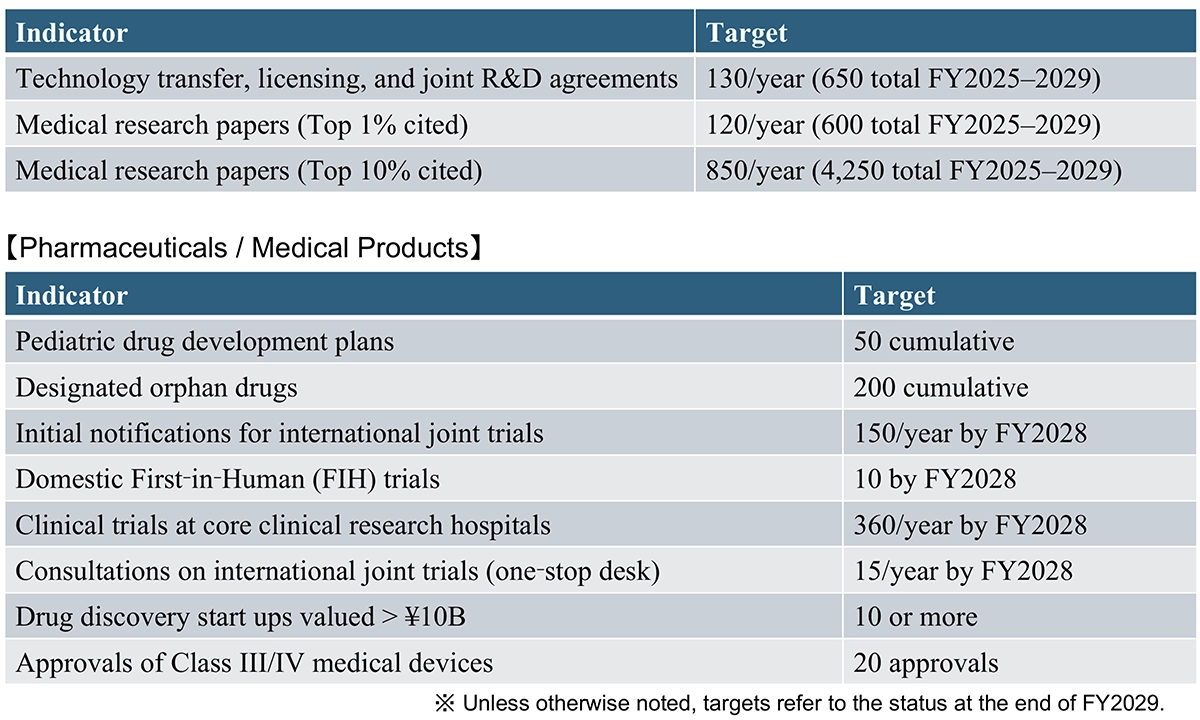
Additionally, new schedules and roadmaps for achieving output indicators have been prepared and issued, and progress will be measured and managed through councils organized by the Cabinet Office. This is expected to ensure the effectiveness of the Strategy more than ever before.
Infectious Disease Countermeasures and Overcoming “Panic and Neglect”
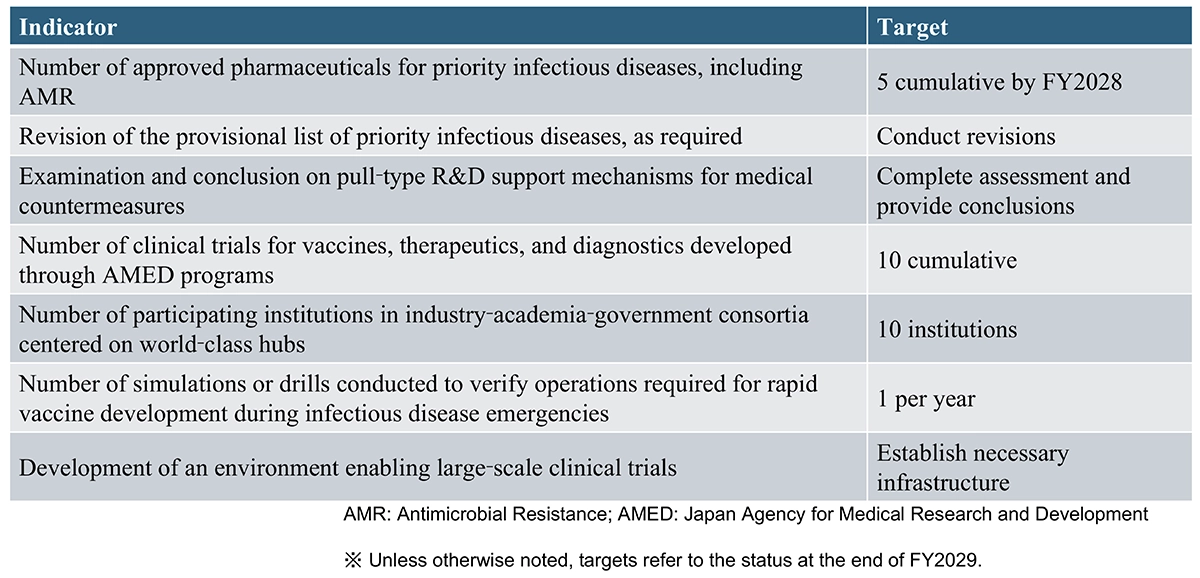
The newly added integrated project on “Infectious Diseases” is a section to which the author devoted particular effort (see Table 2). In Japan, there is a tendency referred to as “Panic and Neglect,” whereby society reacts with great alarm during a pandemic but loses interest once the situation subsides, often leading to criticism of the safety of vaccines developed in response to that pandemic. For this reason, the Strategy explicitly emphasizes not only vaccines but also the promotion of the development of therapeutic drugs and diagnostic tools. In addition to the project for establishing world-class research and development hubs for vaccines, it is expected that similar initiatives for therapeutic drugs and diagnostic tools will be promptly advanced.
Key Measures in the Field of Clinical Trials Under the Third Strategy
To establish a robust drug discovery ecosystem, it is necessary to strengthen all stages of the drug development process. However, considering the importance of improving public access to pharmaceuticals, refining the “exit stage”—more specifically, the reinforcement of clinical trial and regulatory review systems while ensuring equitable access—has become an urgent priority. Accordingly, the Third Strategy is filled with measures related to clinical trials and regulatory review.
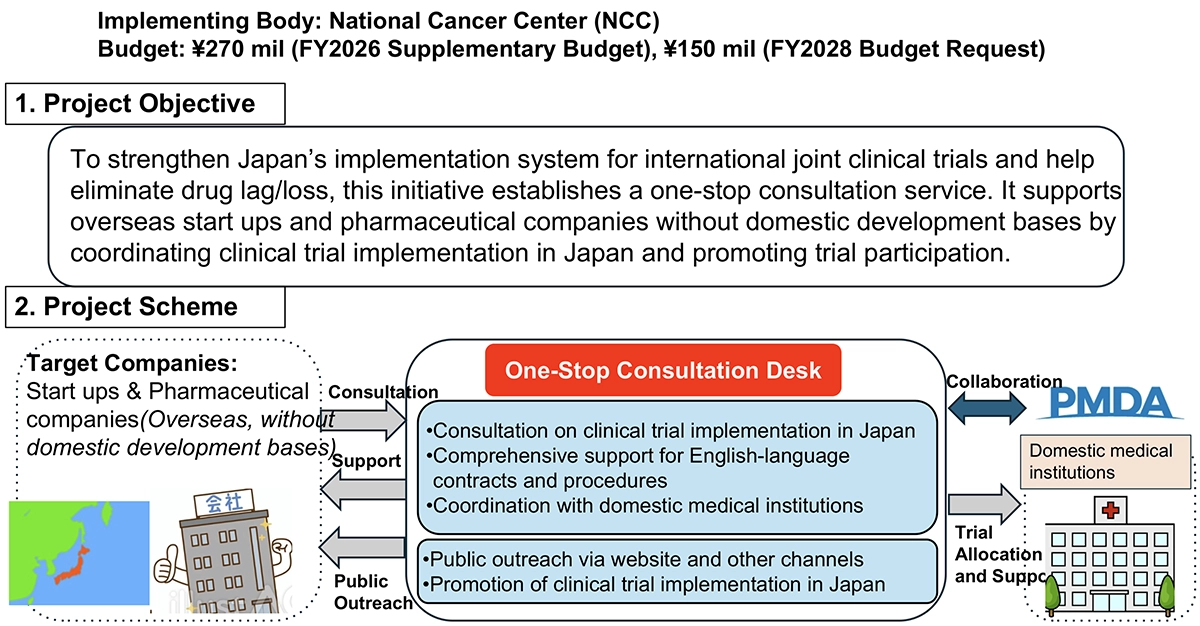
Among them, the One-Stop Consultation Service for International Collaborative Clinical Trials (see Figure 1) is regarded as a particularly important initiative. In the United States (US), start ups are the main drivers of new drug development. Japan, by establishing a consultation service for such companies and facilitating their matching with domestic medical institutions, expects a new form of industry–academia collaboration between overseas companies and Japanese academia. US companies, especially start ups and venture firms, are strongly encouraged to actively utilize this new mechanism for planning their clinical trials in Japan.
Digital transformation (DX) and artificial intelligence (AI) are also crucial. Building upon discussions of AI utilization in basic research, it is now necessary to promote AI use across the entire clinical trial process. The Pharmaceuticals and Medical Devices Agency (PMDA) has also declared its commitment to fully engage in AI utilization, signaling that industry, government, and academia have entered a stage where genuine collaboration is possible.
As part of the follow-up activities for the Third Strategy, approximately 12 billion yen (US$75 million) was allocated in the FY2025 supplementary budget to the Global R&D Support Project for Treatments of Intractable and Rare Diseases. This initiative supports Japan-led international collaborative clinical trials conducted by companies developing innovative pharmaceuticals for intractable and rare diseases, with the aim of eliminating drug lag and drug loss. Furthermore, through the implementation of international collaborative trials, Japan seeks to enhance its clinical development capacity (including trial sites and CROs), foster exchanges among clinical development professionals, and establish a clinical trial system that meets international standards. The FY2024 supplementary budget also increased funding for the development of first-in-human (FIH) research facilities, further strengthening Japan’s clinical trial infrastructure.
Conclusion and Future Opportunities
Prime Minister Sanae Takaichi has long demonstrated a deep understanding of research and development since her service as Minister in charge of the Health and Medical Strategy, and “Drug Discovery and Advanced Medical Fields” have been designated as priority areas in her administration. With favorable winds now supporting R&D in drug discovery, Japan has a new opportunity to advance research and development so that Japanese science can flourish at the center of the global life sciences stage.
Crossing Boundaries, Shaping a Future Beyond Imagination with Innovation in Tokyo.

